��Ŀ����
����Ŀ�����⻯��(NaBH4����Ϊ+3��)Ϊ��ɫ��ĩ���ڸ���������ȶ����ڳ�ʪ�����зֽ⣬�dz��õĻ�ԭ����ƫ�����ƣ�NaBO2��������ˮ���������Ҵ�����ˮ�⡣Ŀǰ�ж��ֹ��տ��Ʊ�NaBH4��
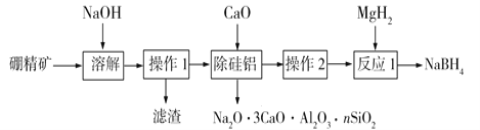
��1��������һ����B2O3����Al2O3��SiO2��Fe2O3�����ʣ���ȡNaBH4���������£�
�١��ܽ���ʱ��B2O3��NaOH��Ӧ������NaBO2����Ӧ���ӷ���ʽΪ____��
�ڡ����������������CaO����������CaCl2��ԭ���У��ܽ��衢���Գ�����ȥ�������������������ӣ�___��
�ۡ�����2���ǽ���Һ�������ᾧ��ϴ�ӣ�����ϴ��ѡ�õ��Լ������_____������ĸ����
a. ��ˮ���� b. �Ҵ��� c. ��ˮ���� d. NaOH��Һ
������Ӧ1����MgH2��NaBO2��ϵõ�NaBH4��MgO���仯ѧ����ʽΪ________��
��2���ҹ�����ƽ������ʴ�缫���ϣ��������ӽ���ĤΪ����Ĥ�����ƫ�����Ƶļ���Һ��Ҳ���Ը�Ч�Ʊ�NaBH4���ù�����������Ϊ________�������缫����ʽΪ_____��
���𰸡�B2O3+2OH- =2BO2-+H2O ʹ��Һ�ʼ��ԣ�����NaBO2 ��ˮ�� b 2MgH2+NaBO2 =NaBH4+2MgO O2 BO2-+8e-+6H2O=BH4-+8OH-
��������
��1���������м����������Ʒ�����Ӧ��B2O3+2NaOH =2NaBO2+H2O��Al2O3+2NaOH=2NaAlO2+H2O��SiO2+2NaOH=Na2SiO3+H2O�����ˡ���������Fe2O3����Һ�м���CaO������Na2O��3CaO��Al2O3��nSiO2����,���˳���������ͬʱ���Ի�������NaBO2��ˮ�⣬��Һ�������ᾧ��ϴ�ӣ��õ�NaBO2��NaBO2��MgH2��Ӧ����NaBH4��
NaBO2��MgH2����NaBH4������þ������
��2�����ݵ�������������ʧ���ӣ�������ԭ�õ��ӣ��ж����������д�缫��Ӧʽ��
��1����B2O3��NaOH��Ӧ������NaBO2��ˮ����Ӧ���ӷ���ʽΪB2O3+2OH- =2BO2-+H2O��
�𰸣�B2O3+2OH- =2BO2-+H2O
�������������������CaO��CaO+H2O=Ca(OH)2,��Һ�ʼ��ԣ���������NaBO2 ��ˮ�⣻
�𰸣�ʹ��Һ�ʼ��ԣ�����NaBO2 ��ˮ��
��������2���ǽ���Һ�������ᾧ��ϴ�ӣ��õ�NaBO2����Ϊƫ�����ƣ�NaBO2��������ˮ���������Ҵ�����ˮ�⣬����ϴ��ѡ�õ��Լ�������Ҵ�����ѡb��
�𰸣�b
������Ӧ1����MgH2��NaBO2��ϵõ�NaBH4��MgO�����������غ�ɵã���ѧ����ʽΪ2MgH2+NaBO2 =NaBH4+2MgO��
�𰸣�2MgH2+NaBO2 =NaBH4+2MgO
��2�����ƫ�����Ƶļ���Һ��������8OH--8e-=4H2O+2O2��������BO2-+8e-+6H2O=BH4-+8OH-��
�𰸣�BO2-+8e-+6H2O=BH4-+8OH-

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�