��Ŀ����
ij��Һ�п��ܺ���Ba2+��Na+��SO42-��CO32-��HCO3-��Cl-�������е�һ�ֻ��֡��ֽ�������ʵ�飺
��ȡ������Һ�����������BaCl2��Һ��������ɫ���������ˣ�������м�����������ᣬ������ȫ�ܽ⣬���������壻
����ʵ��ٵ���Һ�е���AgNO3��Һ�����ְ�ɫ������������ʵ���жϣ�����Һ�п϶����ڵ�������______________________________��
�϶������ڵ�����Ϊ__________ _________������ȷ���Ƿ���ڵ�����________________________��
��1��CO32�D Na�� ��2�֣�
��2��SO42�D Ba2�� ��2�֣�
��3��Cl�D HCO3�D��2�֣�
������������� ��ȡ������Һ�����������BaCl2��Һ��������ɫ���������ܺ���BaSO4,BaCO3�����ˣ�������м�����������ᣬ������ȫ�ܽ⣬���������壻˵��û��BaSO4��һ������CO32�D,ԭ��Һ��һ������SO42�D��Ba2�� ,һ������CO32�D����Һ���������Ӿ��������ӣ��ض�����Na�� ��
����ʵ��ٵ���Һ�е���AgNO3��Һ�����ְ�ɫ���������ڢ��м�����BaCl2����ȷ���Ƿ���Cl�D �����⣬HCO3�DҲ��ȷ���Ƿ���ڡ�
�𰸣���1��CO32�D Na�� ��2�֣�
��2��SO42�D Ba2�� ��2�֣�
��3��Cl�D HCO3�D��2�֣�
���㣺���������ӵļ��飻���ӹ�������

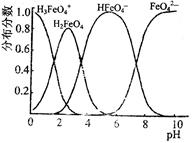
ij�����Һ�п��ܺ��е��������±���ʾ��
| ���ܴ������е������� | H+��Ag+��Mg2+��Al3+��NH ��Fe3+ ��Fe3+ |
| ���ܴ������е������� | Cl-��Br-��I-��CO ��AlO ��AlO |
Ϊ̽����ɷ֣�����������̽��ʵ�顣
��1��̽��һ��
��ͬѧȡһ�����Ļ����Һ����������μ�������������Һ���������������ʵ���(n)��������� ������Һ�������V���Ĺ�ϵ��ͼ��ʾ��
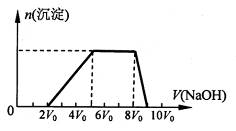
�ٸ���Һ��һ�����е���������______________�����Ӧ���ʵ���Ũ��֮��Ϊ________��һ�������ڵ���������_____________��
����д���������ٹ����з�����Ӧ�����ӷ���ʽ_____________________________��
��2��̽������
��ͬѧ������Һ�к��д�����Cl-��Br-��I-������1 L�û����Һ��ͨ��һ������Cl2����Һ��Cl-��Br-��I-�����ʵ�����ͨ��Cl2���������״�����Ĺ�ϵ���±���ʾ�� ������ش��������⣺
| Cl2���������״���� | 5.6 L | 11.2 L | 22.4 L |
| n (Cl-) | 2.5 mol | 3.0 mol | 4.0 mol |
| n (Br-) | 3.0 mol | 2.8 mol | 1.8 mol |
| n (I-) | x mol | 0 | 0 |
�ٵ�ͨ��Cl2�����Ϊ5.6 Lʱ����Һ�з�����Ӧ�����ӷ���ʽΪ_______________��
��ԭ��Һ��Cl-��Br-��I-�����ʵ���Ũ��֮��Ϊ______________________��
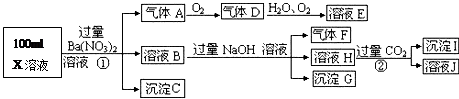
ij��ɫ��Һ�������п��ܴ����������ӣ�Na+��Ag+��Ba2+��Al3+��AlO2-��S2-��CO32-��SO32-��SO42-����ȡ����Һ�����й����飬�������£�
�ش��������⣺
��1�����ɳ��������ӷ���ʽ�� ��
��֪�������������������ɣ�������ˮ������HBr�����������ʵ����һ���������ֳɷ֣��������Լ��Լ����������±��У��ɲ�����������ÿһ�ж�Ӧ��ȷ���ɵ÷֣�
| ���� | ʵ��Ŀ�� | �Լ� | ���� |
| 1 | | | |
| 2 | | | |
| 3 | | | |
| 4 | | | |
��2������Һ�����ɳ����ҵ����ӷ���ʽ�� ��
��3����������������Һ�п϶����ڵ������У� ��
��4�������Һ�п��ܴ��ڵ����ӣ���ʵ����֤���Ƿ���ڵķ�����
��
�Թ��ڱڵ���ǿ������ȵ�KOH��Һ��ϴ�ӣ�3S+6KOH==K2SO3+2K2S+3H2O���÷�Ӧ�У��������뱻��ԭ����ԭ�Ӹ�����Ϊ
| A��1��2 | B��2��1 | C��1��1 | D��3��2 |
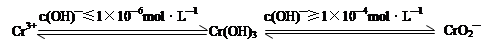


 4Fe(OH)3����8OH��+3O2��,��K2FeO4������ˮ�����е������� ��
4Fe(OH)3����8OH��+3O2��,��K2FeO4������ˮ�����е������� ��