��Ŀ����
ij�����Һ�п��ܺ��е��������±���ʾ��
| ���ܴ������е������� | H+��Ag+��Mg2+��Al3+��NH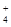 ��Fe3+ ��Fe3+ |
| ���ܴ������е������� | Cl-��Br-��I-��CO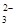 ��AlO ��AlO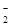 |
Ϊ̽����ɷ֣�����������̽��ʵ�顣
��1��̽��һ��
��ͬѧȡһ�����Ļ����Һ����������μ�������������Һ���������������ʵ���(n)��������� ������Һ�������V���Ĺ�ϵ��ͼ��ʾ��

�ٸ���Һ��һ�����е���������______________�����Ӧ���ʵ���Ũ��֮��Ϊ________��һ�������ڵ���������_____________��
����д���������ٹ����з�����Ӧ�����ӷ���ʽ_____________________________��
��2��̽������
��ͬѧ������Һ�к��д�����Cl-��Br-��I-������1 L�û����Һ��ͨ��һ������Cl2����Һ��Cl-��Br-��I-�����ʵ�����ͨ��Cl2���������״�����Ĺ�ϵ���±���ʾ�� ������ش��������⣺
| Cl2���������״���� | 5.6 L | 11.2 L | 22.4 L |
| n (Cl-) | 2.5 mol | 3.0 mol | 4.0 mol |
| n (Br-) | 3.0 mol | 2.8 mol | 1.8 mol |
| n (I-) | x mol | 0 | 0 |
�ٵ�ͨ��Cl2�����Ϊ5.6 Lʱ����Һ�з�����Ӧ�����ӷ���ʽΪ_______________��
��ԭ��Һ��Cl-��Br-��I-�����ʵ���Ũ��֮��Ϊ______________________��
(1)��H+��NH4+��Al3+��2��3:1��CO32?��AlO2-
��Al(OH)3+OH?= AlO2-+2H2O
(2)Cl2+2I?=I2+2Cl?;10:15:4
���������������1���ٸ���ͼ��NaOH��Һ��������0-2V0ʱ����H+��Ӧ��2V0-4V0ʱ,NaOH��Al3+��Ӧ������������������6V0-8V0ʱ,����������NH4+��Ӧ���������ӷ���ʽ��������ʵ���Ũ��֮��Ϊ2:3:1����ΪAl3+��CO32?��AlO2-��Ӧ�����ܹ��棬�������������Ӳ����ڡ��ڳ�������ʱ�����ķ�ӦΪNaOH��Al(OH)3�ķ�Ӧ��
��2���ٸ��ݻ�ԭ��ǿ����Cl2������I?��Ȼ��������Br?��ͨ��Cl2�����Ϊ5.6 Lʱ��I?����ʣ�࣬Cl2ֻ����I?����ԭ��Һ�е�Cl?Ϊ��2.5mol-5.6L��22.4L/mol��2=2mol��n (Br?)=3.0mol������Cl211.2Lʱ�����������е�I?��0.2mol��Br?��n(I?)=2��(11.2L��22.4L/mol-1/2��0.2mol)=0.8mol,��ˣ�Cl-��Br-��I-�����ʵ���Ũ��֮��Ϊ10��15��4
���㣺���⿼�����ӵ��ƶϡ��������ʵ����ļ��㡢���ӷ���ʽ����д��������ԭ��Ӧ���Ⱥ�˳����ؼ��㡣

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���ԭ�ӷ�����(CN)2������(SCN)2��(OCN)2��������±�ص��ʣ�Cl2��Br2�ȣ����ƣ��ʳ�����Ϊ��±�ء����ǿ�����������Σ����±�������X����F��Cl��Br��I����
| | ±�� | �� | ���� | �� |
| �����ʡ� | X2[ | (CN)2 | (SCN)2 | (OCN)2 |
| �� | HX | HCN | �� | HOCN |
| �� | KX | KCN | KSCN | KOCN |
��1���ڱ��Т٢ڿո�Ӧ�ֱ���д_______��_______��
��2����(CN)2��KOH��Һ��Ӧ�Ļ�ѧ����ʽΪ��__________________________��
����֪�����ӵĻ�ԭ��ǿ��Ϊ��Cl����Br����CN����SCN����I������д����NaBr��KSCN�Ļ����Һ�м���(CN)2��Ӧ�����ӷ���ʽ��____________________________________��
��1������
| ���ʣ����ʣ� | ��ȥ���� �����Լ��� | ���ӷ���ʽ ���������ӷ���ʽ����д��ѧ����ʽ�� |
| Fe(Al) | | |
| SiO2��CaCO3�� | | |
| CO2(HCl) | | |
| NaHCO3��Һ��Na2CO3�� | | |
| Na2CO3��NaHCO3�� | | |
| Al2O3��Al(OH)3�� | | |
��2������������ͨ�뵽�Ȼ�����Һ��δ��������������ͨ�������Һ�еμ���ˮ��ˮ����������ɫ�����������ó����ijɷֱַ�Ϊ �� ����ֱ�д��������Ӧ�����ӷ���ʽ �� ��
��ij��Һ�н����±������е�5�����ӣ�������ˮ�ĵ��뼰���ӵ�ˮ�⣩�������ӵ����ʵ�����Ϊ1mol��
| ������ | SO42-��NO3-��Cl- |
| ������ | Fe3+��Fe2+��NH4+��Cu2+��Al3+ |
������ԭ��Һ�м���KSCN��Һ�������Ա仯��������ԭ��Һ�м�����������ᣬ���������ɣ���Һ������������䡣������ԭ��Һ�м���BaCl2��Һ���а�ɫ�������ɡ��Իش���������
��1��������ԭ��Һ�м�����������ᣬ�ټ���KSCN��Һ�������� ��
��2��ԭ��Һ�к��е��������� ��
��3����ԭ��Һ�м������������ᣬ������Ӧ�����ӷ���ʽΪ ��
��4����ԭ��Һ�м���������NaOH��Һ����ַ�Ӧ���ˡ�ϴ�ӡ����գ��������ù�����������ƽ��������Ϊ ��
��. �����������壨FeC2O4��2H2O����̼��﮺Ͷ�������������и��·�Ӧ���Ʊ�﮵�ص��������Ϲ�������ﮣ�Li2FeSiO4��������������������������н������ط������������ͼ��ʾ��TG%��ʾ������������ռԭ��Ʒ�������İٷ�����,��ش��������⣺
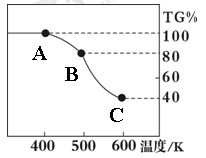
��5����������������̼Ԫ�صĻ��ϼ�Ϊ��
��6��A��B������Ӧ�Ļ�ѧ����ʽΪ ��
��7����ȷ�о�������B��Cʵ���Ƿ��������еģ�ÿһ��ֻ�ͷ�һ�����壬�ڶ����ͷŵ��������Է��������ϵ�һ���Ĵ����һ���ͷŵ����廯ѧʽΪ�� ���ͷŵڶ�������ʱ����Ӧ�Ļ�ѧ����ʽΪ ��
ij�Ͻ�(����ͭ����)��ͭ���������ʵ���֮��Ϊymol������Cu�����ʵ�������Ϊa ������ȫ��Ͷ��50mLbmol��L��1��������Һ�У�����ʹ���ַ�Ӧ(����NO��Ψһ�Ļ�ԭ����)������˵������ȷ����
| A����������ʣ�࣬����Һ���ٵ�����������ֿ�ʼ�ܽ� |
| B��������ȫ���ܽ⣬����Һ�в�һ������Fe3�� |
| C��������ȫ���ܽ⣬�Ҳ���336mL����(��״��)����b=0.3 |
| D������Һ�н�������ֻ��Fe3����Cu2��ʱ����a��b�Ĺ�ϵΪ��b��80y(1��a/3) |