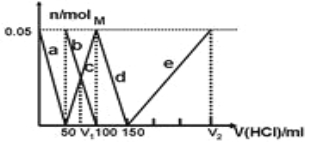
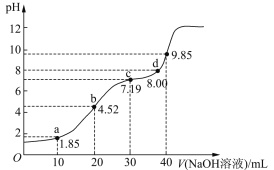
��Ŀ����
����Ŀ����A��B��C��D����ǿ����ʣ�������ˮ�е��������������(ÿ������ֻ��һ���������һ����ظ�)��
������ | Na����Ba2����NH |
������ | CH3COO����OH����Cl����SO |
��֪����A��C��Һ��pH������7��A��B����Һ��ˮ�ĵ���̶���ͬ����C��Һ��D��Һ����ʱֻ���ɰ�ɫ������B��Һ��C��Һ����ʱֻ���ɴ̼�����ζ�����壬A��Һ��D��Һ���ʱ������
(1)A��_____________��B��____________(�ѧʽ) �������ӷ���ʽ��ʾA��ˮ��Һ�д��ڵ�ƽ���ϵ�� ��
(2)25 ��ʱ��0.1 mol��L��1 B��Һ��pH��a����B��Һ�У�c(H��)��c(OH��)��_____________(����Ũ�ȷ���)=_____________(�ú���a�Ĺ�ϵʽ��ʾ)��
(3)��������������ʵ���Ũ�ȵ�B��Һ��C��Һ��ϣ���Ӧ����Һ�и�������Ũ���ɴ�С��˳����_____________��
(4)��һ�������0.005 mol��L��1��C��Һ�У�����һ�������0.00125 mol��L��1�����ᣬ�����Һ��pH��11������Ӧ����Һ���������C��Һ����������֮�ͣ���C��Һ��������������_ _____��
(5)�ڻ�ѧ�����в���K2CrO4Ϊָʾ������AgNO3����Һ�ζ���Һ��Cl������Ag+��CrO42����ש��ɫ������ָʾ����ζ��յ㡣����Һ��Clǡ�ó�����ȫ(Ũ�ȵ���1.0��105 mol��L1)ʱ����Һ��c(Ag+)Ϊ_______ mol��L1����ʱ��Һ��c(CrO42)����__________ mol��L1��(��֪Ag2CrO4��AgCl��Ksp�ֱ�Ϊ2.0��1012��2.0��1010)��
(6)��ʹ������к͵ζ����ⶨ���Ũ�ȡ��ڱ�ʵ��ĵζ������У����в�����ʹʵ����ƫ����� (��д���)��
a����ʽ�ζ����ڵζ�ʱδ�ñ�NaOH��Һ��ϴ
b����ʽ�ζ��ܵļ����ڵζ�ǰ�����ݣ��ζ���������ʧ
c����ƿ�м����������Һ���ټ�����ˮ
d����ƿ�ڵζ�ʱ����ҡ����������Һ�彦��
���𰸡�(1)CH3COONa NH4Cl CH3COO��+H2O![]() CH3COOH+OH�� H2O
CH3COOH+OH�� H2O![]() H++ OH��
H++ OH��
(2)c(NH3��H2O) ��( 10-a��10a��14)mol��L��1
(3)c(OH��)��c(Ba2��)��c(Cl��)��c(NH4+)��c(H��)
(4)1��4 (5)2.0��10-5 5��10-3 (6)ab
��������
�����������A��B��C��D����ǿ����ʣ�������ˮ�е���ʱ�ɲ�����������(ÿ������ֻ����һ���������һ����ظ�)����A��C��ҺpH������7��˵��һ����ǿ�һ��������ǿ���Σ�����ǿ�����Ǵ����ƣ���C��Һ��D��Һ����ֻ���ɰ�ɫ������˵��һ����Һ���б����ӣ�һ����Һ������������ӣ�B��Һ��C��Һ����ʱֻ���ɴ̼�����ζ�����壬˵��һ����Һ����笠����ӣ�һ����Һ�������������ӣ�A��Һ��D��Һ���ʱ��������������ͬŨ�ȵ�A��B����Һ��ˮ�ĵ���̶���ͬ������A�Ǵ����ơ�B���Ȼ�李�C������������D�������ƣ�
(1)ͨ�����Ϸ���֪��A��CH3COONa��B��NH4Cl����CH3COONa��Һ�д���CH3COO����ˮ��ƽ���ˮ�ĵ���ƽ�⣬ˮ�ⷴӦʽΪCH3COO��+H2O![]() CH3COOH+OH����ˮ�ĵ��뷽��ʽΪH2O
CH3COOH+OH����ˮ�ĵ��뷽��ʽΪH2O![]() H++ OH����
H++ OH����
(2)������Һ�е���غ��c(H+)+c(NH4+)=c(OH-)+c(Cl-)��������Һ�������غ�� c(NH3��H2O) + c(NH4+) = c(Cl-)����������ʽ�����c(H+)-c(NH3H2O)=c(OH-)��c(H��)��c(OH��)��c(NH3H2O)=10-a-10a-14��
(3)��������֪���Ȼ�狀��������������ʵ�����ȣ���Ϻ���Һ�е������ǰ�ˮ���Ȼ����������������Ȼ���������������Ũ����ȣ���ˮ��Ũ�����Ȼ�������������Ũ�ȵ�2������Һ������������Ũ��������Ӻ�������Ũ����ȣ���ˮ������ֵ��뵼��������Ũ�ȴ���笠�����Ũ�ȣ���Һ�ʼ��ԣ�������Ũ����С��������Һ������Ũ�ȴ�С˳����c(OH-)��c(Ba2+)=c(Cl-)��c(NH4+)��c(H+)��
(4)�����Һ��pH=11������Һ������������Ũ��=![]() mol/L=10-3 mol/L�����������������ΪX����������ΪY��c(OH-)=
mol/L=10-3 mol/L�����������������ΪX����������ΪY��c(OH-)=![]() =10-3 mol/L��X��Y=1��4��
=10-3 mol/L��X��Y=1��4��
(5)����Һ��Cl-��ȫ����ʱ����c(Cl-)=1.0��10-5mol/L������Ksp(AgCl)=2.0��10-10������õ�c(Ag+)=![]() =2.0��10-5mol/L����ʱ��Һ��c(CrO42-)=
=2.0��10-5mol/L����ʱ��Һ��c(CrO42-)=![]() =5.0��10-3mol/L��
=5.0��10-3mol/L��
(6)a����ʽ�ζ����ڵζ�ʱδ�ñ�NaOH��Һ��ϴ����ҺŨ�Ƚ��ͣ����V(��)ƫ����c(����)=![]() ��������֪c(����)ƫ��a��ȷ��b����ʽ�ζ����ڼ����ڵζ�ǰ�����ݣ��ζ���������ʧ�����V(��)ƫ��c(����)=
��������֪c(����)ƫ��a��ȷ��b����ʽ�ζ����ڼ����ڵζ�ǰ�����ݣ��ζ���������ʧ�����V(��)ƫ��c(����)=![]() ��������֪c(����)ƫ��b��ȷ��c����ƿ�м������״���Һ���ټ�����ˮ����V(��)��Ӱ�죬����c(����)=
��������֪c(����)ƫ��b��ȷ��c����ƿ�м������״���Һ���ټ�����ˮ����V(��)��Ӱ�죬����c(����)=![]() ��������֪c(����)��Ӱ�죬��c����d����ƿ�ڵζ�ʱ����ҡ����������Һ�彦��������Һ���ʵ���ƫС�����V(��)ƫС������c(����)=
��������֪c(����)��Ӱ�죬��c����d����ƿ�ڵζ�ʱ����ҡ����������Һ�彦��������Һ���ʵ���ƫС�����V(��)ƫС������c(����)=![]() ��������֪c(����)ƫС����d��ȷ����ѡab��
��������֪c(����)ƫС����d��ȷ����ѡab��

����Ŀ���±���Ԫ�����ڱ���һ���֣��١������ijԪ�أ���ش��������⣺
���� ���� | IA | IIA | IIIA | IVA | VA | VIA | VIIA | 0 |
�� | �� | �� | �� | |||||
�� | �� | �� | �� | �� | �� | �� | ||
�� | �� |
��1���ڢ١�����,��ѧ��������õ�Ԫ����ԭ�ӽṹʾ��ͼΪ___________��
��2���ڢ١����У���������ǿ��Ԫ����_________(��Ԫ�ط���)���ڻ������о�ֻ�Ը��۵�Ԫ����__________(��Ԫ�ط���)��
��3���ڢܡ����У�Ԫ�ص�����������Ӧ��ˮ������������ǿ����__________(�����ʻ�ѧʽ,��ͬ)��������ǿ����________��
��4���ڢܡ����У�ԭ�Ӱ뾶��С����__________(��Ԫ�ط���)�������Ӱ뾶��С����_______(�����ӷ���)��
��5���ڢ١��ۡ��ߡ���Ԫ�ص���̬�⻯�������ȶ�����__________(���⻯�ﻯѧʽ)��
��6��д����ҵ���âٵ����ڸ���������ȡ�ߵ��ʵĻ�ѧ����ʽ______________________��