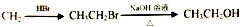��Ŀ����
 ̼���⡢����������ȷǽ���Ԫ�����������������ϢϢ��أ��ش��������⣺
̼���⡢����������ȷǽ���Ԫ�����������������ϢϢ��أ��ش��������⣺��1��д����ԭ�ӵĵ����Ų�ʽ
��2��������ˮ��Һ�д��������
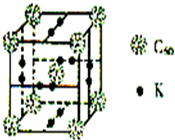
��3����ѧ�Ұ�C60��K������һ��������һ�ָ���ϩ��صĻ�����������ڵ���ʱ��һ�ֳ����壬�侧������ͼ��ʾ����������Kԭ�Ӻ�C60���ӵĸ�����Ϊ
��4����C60��ѧ���ֺϳ���Si60��N60����������������۵㣺Si60��N60��C60�����ƻ���������Ҫ��������N60��C60��Si60����ԭ���ǣ�
��5��Co3+�ж���������Co��CN��63-��Co��NH3��4Cl2+ �ȣ��Rh����������ͬ��Ԫ�أ�ijЩ�������ƣ��������ij�����ΪCsRh��SO4��2?4H2O�����ܽ���ˮ������ˮ��Һ�м���һ��Ũ�ȵ�BaCl2��Һ���������ɣ���д�������ܽ���ˮ��ĵ��뷽��ʽ��
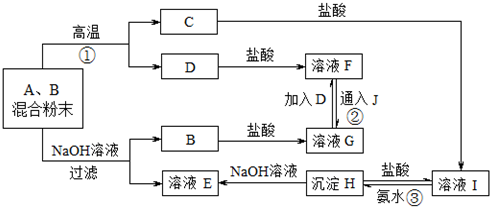
��6������þ�ľ�����
���㣺ԭ�Ӻ�������Ų�,�����ijɼ����,�����ļ���,�������������,��ͬ����Ľṹ��������������������
ר�⣺��ѧ���뾧��ṹ
��������1��SiԪ��ԭ�Ӻ��������Ϊ14�������������ԭ����д��������Ų�ʽ��
ͬ����������ҵ縺������ͬ�������϶��µ縺�Լ�С��
��2��HF����֮�䡢ˮ����֮�䡢HF��ˮ������Hԭ����F��Oԭ��֮�䶼���������
��3�����þ�̯�����㾧����Kԭ�Ӻ�C60���ӵĸ����ȣ�
��4���ṹ���Ƶķ��Ӿ����۵�������Է������������ȣ��ƻ�������Ҫ�Ͽ���ѧ����Ԫ�صĻ�ѧ�����ȶ�����Ԫ�صĵ縺�Գ����ȣ�
��5��CsRh��SO4��2?4H2O�����ܽ���ˮ������ˮ��Һ�м���һ��Ũ�ȵ�BaCl2��Һ���������ɣ�˵�������Ϊ���壬�Rh����������ͬ��Ԫ�أ�ijЩ�������ƣ���Co3+�γɵ�������֪CsRh��SO4��2?4H2O��������ĿΪ6������ΪCs+��[Rh��SO4��2��H2O��4]-��
��6���������������ͬ�����Ӱ뾶ԽС������Խ������Խ��Խ�������ҽ���������
ͬ����������ҵ縺������ͬ�������϶��µ縺�Լ�С��
��2��HF����֮�䡢ˮ����֮�䡢HF��ˮ������Hԭ����F��Oԭ��֮�䶼���������
��3�����þ�̯�����㾧����Kԭ�Ӻ�C60���ӵĸ����ȣ�
��4���ṹ���Ƶķ��Ӿ����۵�������Է������������ȣ��ƻ�������Ҫ�Ͽ���ѧ����Ԫ�صĻ�ѧ�����ȶ�����Ԫ�صĵ縺�Գ����ȣ�
��5��CsRh��SO4��2?4H2O�����ܽ���ˮ������ˮ��Һ�м���һ��Ũ�ȵ�BaCl2��Һ���������ɣ�˵�������Ϊ���壬�Rh����������ͬ��Ԫ�أ�ijЩ�������ƣ���Co3+�γɵ�������֪CsRh��SO4��2?4H2O��������ĿΪ6������ΪCs+��[Rh��SO4��2��H2O��4]-��
��6���������������ͬ�����Ӱ뾶ԽС������Խ������Խ��Խ�������ҽ���������
���
�⣺��1��Si��14��Ԫ�أ���ԭ�Ӻ�����14�����ӣ������������ԭ��֪�����������Ų�ʽΪ��1s22s22p63s23p1��ͬһ������Ԫ�صĵ縺������ԭ�����������������ͬһ������Ԫ�صĵ縺������ԭ���������������С��C��N����ͬһ���ڣ�C��ԭ������С��N������C�ĵ縺��С��N��C��SiԪ������ͬһ���壬C��ԭ������С��SiԪ�أ���CԪ�صĵ縺�Դ���Si�����Ե縺��N��C��Si��
�ʴ�Ϊ��1s22s22p63s23p1��N��C��Si��
��2���������ˮ��Һ�д��ڵ�����У���������Ӽ䡢ˮ���Ӽ䡢ˮ����ԭ�Ӻͷ������з�ԭ��֮�䡢����������ԭ�Ӻ�ˮ����ԭ��֮�䣬����һ����4�������
�ʴ�Ϊ��4��
��3���þ�����C60����=1+8��
=2��6��2��
=6������Kԭ�Ӻ�C60���ӵĸ�����Ϊ6��2=3��1���ʴ�Ϊ��3��1��
��4��Si60��N60��C60���ǽṹ���Ƶķ��Ӿ��壬���Ӿ�����۵�������Է������������ȣ���Է�������Խ���Ӽ�������Խǿ���ۻ���������Խ�࣬�����۵�Si60��N60��C60���ƻ�������Ҫ�Ͽ���ѧ����Ԫ�صĵ縺��Խ�����γɵĻ�ѧ��Խ�ȶ����ϼ���Ҫ������Խ�࣬�����ƻ�������Ҫ������˳��Ϊ��N60��C60��Si60��
�ʴ�Ϊ�����ڽṹ���Ƶķ��Ӿ������Է�������Խ���Ӽ����������»�����Խǿ���ۻ����������Խ�࣬���۵���Si60��N60��C60�����ƻ�������Ͽ���ѧ����Ԫ�ص縺��Խǿ���γɵĻ�ѧ��Խ�ȶ�������ɼ�������Խ�࣬�ɼ�ԭ�Ӱ뾶ԽС���ϼ���������Խ�࣬���ƻ�������Ҫ������˳��ΪN60��C60��Si60��
��5��CsRh��SO4��2?4H2O�����ܽ���ˮ������ˮ��Һ�м���һ��Ũ�ȵ�BaCl2��Һ���������ɣ�˵�������Ϊ���壬�Rh����������ͬ��Ԫ�أ�ijЩ�������ƣ���Co3+�γɵ�������֪CsRh��SO4��2?4H2O��������ĿΪ6������ˮ����ΪCs+��[Rh��SO4��2��H2O��4]-�����뷽��ʽΪ��CsRh��SO4��2?4H2O=Cs++[Rh��SO4��2��H2O��4]-��
�ʴ�Ϊ��CsRh��SO4��2?4H2O=Cs++[Rh��SO4��2��H2O��4]-��
��6������þ�������ƾ������������������ͬ��þ���Ӱ뾶С�ڸ����Ӱ뾶�����Ӱ뾶ԽС������Խ�ʾ����ܣ�����þ�������ƣ�������Խ��Խ�������ҽ�������������þ�����״��ҽ���������
�ʴ�Ϊ�����ڣ�����þ��
�ʴ�Ϊ��1s22s22p63s23p1��N��C��Si��
��2���������ˮ��Һ�д��ڵ�����У���������Ӽ䡢ˮ���Ӽ䡢ˮ����ԭ�Ӻͷ������з�ԭ��֮�䡢����������ԭ�Ӻ�ˮ����ԭ��֮�䣬����һ����4�������
�ʴ�Ϊ��4��
��3���þ�����C60����=1+8��
| 1 |
| 8 |
| 1 |
| 2 |
��4��Si60��N60��C60���ǽṹ���Ƶķ��Ӿ��壬���Ӿ�����۵�������Է������������ȣ���Է�������Խ���Ӽ�������Խǿ���ۻ���������Խ�࣬�����۵�Si60��N60��C60���ƻ�������Ҫ�Ͽ���ѧ����Ԫ�صĵ縺��Խ�����γɵĻ�ѧ��Խ�ȶ����ϼ���Ҫ������Խ�࣬�����ƻ�������Ҫ������˳��Ϊ��N60��C60��Si60��
�ʴ�Ϊ�����ڽṹ���Ƶķ��Ӿ������Է�������Խ���Ӽ����������»�����Խǿ���ۻ����������Խ�࣬���۵���Si60��N60��C60�����ƻ�������Ͽ���ѧ����Ԫ�ص縺��Խǿ���γɵĻ�ѧ��Խ�ȶ�������ɼ�������Խ�࣬�ɼ�ԭ�Ӱ뾶ԽС���ϼ���������Խ�࣬���ƻ�������Ҫ������˳��ΪN60��C60��Si60��
��5��CsRh��SO4��2?4H2O�����ܽ���ˮ������ˮ��Һ�м���һ��Ũ�ȵ�BaCl2��Һ���������ɣ�˵�������Ϊ���壬�Rh����������ͬ��Ԫ�أ�ijЩ�������ƣ���Co3+�γɵ�������֪CsRh��SO4��2?4H2O��������ĿΪ6������ˮ����ΪCs+��[Rh��SO4��2��H2O��4]-�����뷽��ʽΪ��CsRh��SO4��2?4H2O=Cs++[Rh��SO4��2��H2O��4]-��
�ʴ�Ϊ��CsRh��SO4��2?4H2O=Cs++[Rh��SO4��2��H2O��4]-��
��6������þ�������ƾ������������������ͬ��þ���Ӱ뾶С�ڸ����Ӱ뾶�����Ӱ뾶ԽС������Խ�ʾ����ܣ�����þ�������ƣ�������Խ��Խ�������ҽ�������������þ�����״��ҽ���������
�ʴ�Ϊ�����ڣ�����þ��
���������⿼���������Ų����縺�ԡ�������������㡢�������������ʡ������ȣ����dz���֪ʶ�㣬ע��������ڽ�����磬���վ����������۷е��жϣ��Ѷ��еȣ�

��ϰ��ϵ�д�
 ������ѧ���̲���ȫ���ϵ�д�
������ѧ���̲���ȫ���ϵ�д�
�����Ŀ
X��Y��Z��Ϊ������Ԫ�أ������ڱ��е�λ����ͼ��ʾ������������ȷ���ǣ�������
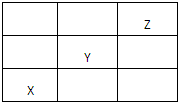

| A��Xһ���ǻ��ý��� |
| B��Y����������������������� |
| C��Z�ĵ�����˫ԭ�ӷ��� |
| D���⻯����ȶ��ԣ�Y��X |
������ʵ����ص�������ȷ���ǣ�������
| A���ñ���ȡ��ˮ�е�Br2ʱ���л���ӷ�Һ©�����¶˷ų� |
| B��������Һ���Ƿ���SO42-�����ε���Ba��NO3��2��Һ��ϡ���� |
| C��������Һʱ������ˮ��������ƿ�̶ȣ�Ӧ�ý�ͷ�ιܽ�������Һ���� |
| D������AlCl3��Һʱ����AlCl3�ܽ��ڽ�Ũ�������У�����ˮϡ�͵�����Ũ�� |
������ˮΪԭ�Ͽ����Ƶ�CO��H2����CH4+H2O?CO+3H2�������й��ڼ�����ˮ��Ӧ��˵������ȷ���ǣ�������
| A��CH4�ȱ������ֱ���ԭ |
| B��H2O�Ȳ����������ֲ��ǻ�ԭ�� |
| C���ڷ�Ӧ�����£�ÿ����1mol H2O��Ȼ����67.2L H2 |
| D������ƽ���ʹ�ø���Ч�Ĵ�������ʹH2�IJ������ |
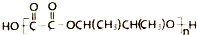 �ĺϳ�·������ͼ�����Լ����ã����ϳ�·������ͼ��ͼ��
�ĺϳ�·������ͼ�����Լ����ã����ϳ�·������ͼ��ͼ��