��Ŀ����
����Ŀ����Ҫ��д�����и��л���Ľṹ��ʽ��
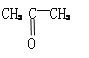
��1����Է�������Ϊ114�������ף��������κ���̼ͬԭ������ϩ����H2�õ�����Ľṹ��ʽΪ____________��
��2����֪������A�뻯����B ��ת����ϵΪ��A![]() B���һ�����B�н���4��̼ԭ�ӡ�1����ԭ�ӡ�1����ԭ�ӣ���B�Ľṹ��ʽΪ____________��
B���һ�����B�н���4��̼ԭ�ӡ�1����ԭ�ӡ�1����ԭ�ӣ���B�Ľṹ��ʽΪ____________��
��3��ij��B����ʽΪCnH2n��2��������KMnO4��Һ������õ���
 ��
�� ��
��
������Ľṹ��ʽΪ____________��
��4��ij��A����Է�������Ϊ84���ú�����ײ�֪�����к���̼̼˫�����ú˴Ź������������ʾֻ��һ�����͵���ԭ�ӡ���A�Ľṹ��ʽΪ____________��
���𰸡���1��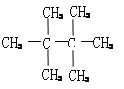 ����2��
����2��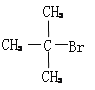 ����3��
����3�� ����4��
����4��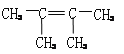 ��
��
�������������������1������������ͨʽ���������ķ���ʽΪC8H18��������ϩ�������������ӳɷ�Ӧ���ɣ�˵���������̼ԭ��������ԭ�ӣ���˽ṹ��ʽΪ�� ����2����4��̼ԭ�ӵ���������ṹ��ʽΪ��CH3CH2CH2CH3��(CH3)3CH��ֻ��1����ԭ�Ӻ�1����ԭ�ӣ����B�Ľṹ��ʽΪ��
����2����4��̼ԭ�ӵ���������ṹ��ʽΪ��CH3CH2CH2CH3��(CH3)3CH��ֻ��1����ԭ�Ӻ�1����ԭ�ӣ����B�Ľṹ��ʽΪ�� ����3������̼̼˫�������������Һ������ʵ�ʣ�����ʻ��е�̼����һ���л������ʻ���̼��̼̼˫������ʽ���ӣ������ṹ��ʽΪ��
����3������̼̼˫�������������Һ������ʵ�ʣ�����ʻ��е�̼����һ���л������ʻ���̼��̼̼˫������ʽ���ӣ������ṹ��ʽΪ�� ����4����Ϊ����̼̼˫��������ϩ������A����ʽΪC6H12��ֻ��һ����ԭ�ӣ�˵���ǶԳƽṹ�����A�Ľṹ��ʽΪ��
����4����Ϊ����̼̼˫��������ϩ������A����ʽΪC6H12��ֻ��һ����ԭ�ӣ�˵���ǶԳƽṹ�����A�Ľṹ��ʽΪ�� ��
��

 ���б�ˢ��ϵ�д�
���б�ˢ��ϵ�д�����Ŀ�������£���ijһԪ��HA��Һ��NaOH��Һ�������ϣ�������Һ��Ũ�Ⱥͻ�Ϻ�������Һ��pH���±���
ʵ���� | HA���ʵ���Ũ����mol��L��1�� | NaOH���ʵ���Ũ����mol��L��1�� | �����Һ��pH |
�� | 0.1 | 0.1 | pH��9 |
�� | c | 0.2 | pH��7 |
�� | 0.2 | 0.1 | pH<7 |
��1���ڢ���ʵ����0.1 mol��L��1HA��Һ��ˮ�ĵ���̶�Ϊa��0.1 mol��L��1NaOH��Һ��ˮ�ĵ���̶�Ϊb�����û����Һ��ˮ�ĵ���̶�Ϊc����a��b��c�ɴ�С��˳����_________��
��2���ڢ���ʵ����c_____0.2���<����������������
��3�����ݢ���ʵ�����õĻ����Һ������Һ������Ũ�ȴ�С����˳����____________��
��4�������£���֪KSP[Cu��OH��2]��2��10��20��ijCuSO4��Һ��c��Cu2������0.02 mol��L��1�����Ҫ����Cu��OH��2��������Ӧ������ҺpH����_________��