��Ŀ����
6��ʵ���ǽ����ѧ����Ļ���;���������й�ʵ�����������ȷ���ǣ�������| A�� | 240 mL 0.2 mol•L-1��NaOH��Һ���ƣ���Ҫʹ����ƽ���ձ���250 mL����ƿ������ | |
| B�� | ��ȥCu���е�CuO�����������еμ�����ϡ�����ٹ��� | |
| C�� | Ϊ��ǿ��ˮ��Ư���ԣ������м���̼��� | |
| D�� | Ϊʹ0.5mol/L��NaNO3��Һ���ʵ���Ũ�ȱ�Ϊ1mol/L���ɽ�ԭ��Һ����������50��ˮ |
���� A������240 mL 0.2 mol•L-1NaCl��Һ����Ϊ�����㡢�������ܽ⡢��ȴ��ת�ơ�ϴ�ӡ����ݡ�ҡ�ȵȣ��������Ʋ���ѡ��ʹ�õ�������
B��CuO�����ᷴӦ����Cu���ܣ�
C�������̼��Ʒ�Ӧ�����Ȼ��ƣ��ٽ��������ܽ⣻
D��100mL��Һ������50gˮ��ʣ����Һ���δ֪�����ܼ������ʵ���Ũ�ȣ�
��� �⣺A������240 mL 0.2 mol•L-1NaCl��Һʹ�õ������У�������ƽ��ҩ�ס��ձ�����������250mL����ƿ����ͷ�ιܣ���A��ȷ��
B��CuO�����ᷴӦ����Cu���ܣ����������еμ�����ϡ������ܳ��ӣ���B��ȷ��
C��Cl2+H2O?HCl+HClO�������̼��Ʒ�Ӧ�����Ȼ��ơ�������̼��ˮ���Ӷ��ٽ������ܽ⣬������Һ�ʴ�����Ũ����������Һ��Ư������ǿ����C��ȷ��
D��������50gˮ��ʣ����Һδ�����ܶȣ���������Һ���������D����
��ѡD��
���� ���⿼�黯ѧʵ�鷽�������ۣ�Ϊ��Ƶ���㣬�漰��Һ���ơ����������ᴿ����ѧƽ���ƶ��ȣ��ۺ��Խ�ǿ���������ʼ���Ӧԭ���Ŀ��飬ע��ʵ��������Է�������Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
17�����úϳ�������Ҫ�ɷ�Ϊ CO��CO2�� H2���ڴ����������ºϳɼ״�������������Ӧ���£�
��CO��g��+2H2��g��?CH3OH��g����H1=-99kJ?mol-1
��CO2��g��+3H2��g��?CH3OH��g��+H2O��g����H2
��CO2��g��+H2��g��?CO��g��+H2O��g����H3=+41kJ?mol-1
��֪��Ӧ���е���صĻ�ѧ�������������£��� C-H ���ܺ͡�H2�ֱ��ǣ�������
��CO��g��+2H2��g��?CH3OH��g����H1=-99kJ?mol-1
��CO2��g��+3H2��g��?CH3OH��g��+H2O��g����H2
��CO2��g��+H2��g��?CO��g��+H2O��g����H3=+41kJ?mol-1
��֪��Ӧ���е���صĻ�ѧ�������������£��� C-H ���ܺ͡�H2�ֱ��ǣ�������
| ��ѧ�� | H-H | C-O | C�TO | H-O | C-H |
| E/��kJ��mol-1�� | 436 | 343 | 1076 | 465 | 413 |
| A�� | 403 kJ•mol-1��-66 kJ•mol-1 | B�� | 423 kJ•mol-1��+28 kJ•mol-1 | ||
| C�� | 314 kJ•mol-1��-18 kJ•mol-1 | D�� | 413 kJ•mol-1��-58 kJ•mol-1 |
14�����ж�ʵ���������������ǣ�������
| A�� | ����0.1mol/L NaCl��Һʱ����û��ϴ���ձ��Ͳ���������������Һ���ʵ���Ũ��ƫ�� | |
| B�� | NH4NO3�ܽ����ȣ�������0.5mol/L H4NO3��Һʱֱ�ӽ��ܽ�����Һת�Ƶ�����ƿ�У���������Һ�����ʵ���Ũ��ƫ�� | |
| C�� | ����һ�����ʵ���Ũ����Һʱ�������������Ѿ����⣬��������Һ�����ʵ���Ũ��ƫ�� | |
| D�� | ����һ�����ʵ���Ũ����Һʱ���������в�С�ļ�ˮ�����̶��ߣ����̽�������ˮ��������������Һ�����ʵ���Ũ��ƫ�� |
1������˵������ȷ���ǣ�������
| A�� | Ħ���������ʵ����ĵ�λ�������ʵ�������λ | |
| B�� | ���ʵ����ȱ�ʾ���ʵ������ֱ�ʾ���ʵ����� | |
| C�� | �����ӵ�������12kg̼12�к��е�̼ԭ����Ŀ | |
| D�� | 1molH2O���10mol���� |
11�������أ�NiMH��Ŀǰ�Ѿ���Ϊ��϶���������һ����Ҫ������ͣ�NiMH�е�M��ʾ���������Ͻ𣮸õ���ڳ������е��ܷ�Ӧ����ʽ�ǣ�Ni��OH��2+M=NiOOH+MH����֪��6NiOOH+NH3+H2O+OH-=6Ni��OH��2+NO2- ����˵����ȷ���ǣ�������
| A�� | ��������OH-���Ӵ�����������Ǩ�� | |
| B�� | �������������ĵ缫��Ӧʽ��H2O+M+e-=MH+OH-��H2O�е�H��M��ԭ | |
| C�� | ��طŵ�����У������缫��ӦʽΪ��NiOOH+H2O+e-=Ni��OH��2+OH- | |
| D�� | NiMH����п�����KOH��Һ����ˮ����Ϊ�������Һ |
16�����ж�������ԭ��Ӧ�ķ�����ȷ���ǣ�������
| A�� |  | B�� |  | ||
| C�� | 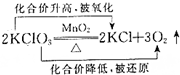 | D�� |  |