��Ŀ����
����Ŀ���������Ƶ��������ס����ӡ��ɳ������̫���ܣ���������Чת��Ϊ����Դ�������ӡ�����ں����ֱ������Ӻ���п���ֲ�ͬ�İ뵼�������ɡ�
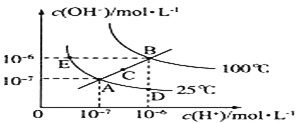
��1����֪Cd��Znλ��ͬһ���壬����Zn����һ���ڣ���Cd�ļ۵����Ų�ͼΪ___����֪Zn��Cu�������仯��ͼ��ʾ���Խ��Ͳ���������������ڲ���ܵ�ԭ��___��

��2��CdSȼ�տ�����CdO��SO2��SO2�ڿ������������Ỻ��ת��ΪSO3��SO2��S���ӻ���ʽΪ___��SO3���ӵĿռ乹��Ϊ___��
��3��O��S��SeΪͬ����Ԫ�أ���֪���Ӧ�⻯�������������±���

��H2Se�е����H2S��ԭ��Ϊ___��
��H2O�ķֽ��¶ȸ���H2S��ԭ��Ϊ___��
��4����֪ZnS�۵�Ϊ2830�棻CdS�۵�Ϊ1750�棬���������___���壬ZnS�۵���ߵ�ԭ��Ϊ___��
��5����ͼΪZnS�ľ���ͼ������֪�����п����֮�����Ϊapm����ZnS������ܶ�Ϊ___g��cm-3���г�����ʽ���ɣ���

���𰸡�![]() Cu+��3d���Ϊȫ���ṹ�������ͣ����ȶ� sp2 ƽ�������� H2Se��H2S��Ϊ���Ӿ��壬H2S���ԭ������С�����Ӽ�������С���е�� ԭ�Ӱ뾶��O��S���ʹ��ۼ�O-H���ļ���С��S-H�������ܣ�O-H��S-H�����H2O���ȶ��Ը���H2S���ֽ��¶ȸ��ߣ���O-H����S-H���ȶ���H2O�ķֽ��¶ȱ�H2S���ߣ������������𰸣� ���� �������Ӱ뾶Cd2+����Zn2+���ʾ�����ZnS��CdS�����۵�ZnS��CdS
Cu+��3d���Ϊȫ���ṹ�������ͣ����ȶ� sp2 ƽ�������� H2Se��H2S��Ϊ���Ӿ��壬H2S���ԭ������С�����Ӽ�������С���е�� ԭ�Ӱ뾶��O��S���ʹ��ۼ�O-H���ļ���С��S-H�������ܣ�O-H��S-H�����H2O���ȶ��Ը���H2S���ֽ��¶ȸ��ߣ���O-H����S-H���ȶ���H2O�ķֽ��¶ȱ�H2S���ߣ������������𰸣� ���� �������Ӱ뾶Cd2+����Zn2+���ʾ�����ZnS��CdS�����۵�ZnS��CdS 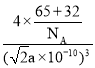
��������
(1)Zn�ļ۲�����Ų�ʽΪ3d104s2������Zn��Cd�����ڱ���λ�ù�ϵ������ȫ��״̬ʱ�ṹ�ȶ�����ʧȥһ������ʱ��Ҫ���������ࣻ
(2)��������ԭ�Ӽ۵��ӶԻ������۷����ӻ����ͺͿռ乹�ͣ�
(3)�ٷ��Ӿ������Է�������Խ���۷е�Խ�ߣ�
�ڰ뾶ԽС������Խ�̣�����Խ�������
(4)�����۷е��жϾ������ͣ����Ӱ뾶ԽС��������Խ���۷е�Խ�ߣ�
(5)���ݾ����Ľṹ�������ܶȹ�ʽ���㡣
(1)����Zn�ļ۲�����Ų�ʽΪ3d104s2���ɼ���Znͬһ��������Zn��һ���ڵ�Cd�ļ۵����Ų�Ϊ4d105s2���۵����Ų�ͼΪ��![]() ���������仯ͼ��֪I1(Cu)��I1(Zn)��I2(Cu)��I2(Zn)������Cu+��3d���Ϊȫ���ṹ�������ͣ����ȶ�����Zn+��4s�����һ�����ӣ�Cu+��ʧȥһ������ʱ��Ҫ���������ࣻ
���������仯ͼ��֪I1(Cu)��I1(Zn)��I2(Cu)��I2(Zn)������Cu+��3d���Ϊȫ���ṹ�������ͣ����ȶ�����Zn+��4s�����һ�����ӣ�Cu+��ʧȥһ������ʱ��Ҫ���������ࣻ
(2)SO2��S�ĵļ۲���Ӷ���=2+![]() (6-2��2)=3��������ԭ�Ӳ���sp2�ӻ���SO3��S�ĵļ۲���Ӷ���=3+
(6-2��2)=3��������ԭ�Ӳ���sp2�ӻ���SO3��S�ĵļ۲���Ӷ���=3+![]() (6-3��2)=3��������ԭ�Ӳ���sp2�ӻ����ռ乹��Ϊƽ�������Σ�
(6-3��2)=3��������ԭ�Ӳ���sp2�ӻ����ռ乹��Ϊƽ�������Σ�
(3)��H2Se��H2S��Ϊ���Ӿ��壬���ڷ��Ӿ������ԭ������H2Se��H2S���ʷ��Ӽ�������H2Se��H2S����˷е�H2Se��H2S��
������ԭ�Ӱ뾶��O��S���ʹ��ۼ�O-H���ļ���С��S-H�������ܣ�O-H��S-H�����H2O���ȶ��Ը���H2S����ֽ��¶ȸ���H2S��
(4)����ZnS�۵�Ϊ2830����CdS�۵�Ϊ1750�������Ƚϸߣ��ʶ��������Ӿ��壻�������Ӱ뾶Cd2+����Zn2+���ʾ�����ZnS��CdS�����۵�ZnS��CdS��
(5)���������Znԭ�Ӽ�ľ��루������������֮��ľ��룩Ϊapm������Խ���Ϊ2apm�����ı߳�Ϊ![]() apm=
apm=![]() a��10-10cm���ʾ��������Ϊ(
a��10-10cm���ʾ��������Ϊ(![]() a��10-10)3cm3����������Znԭ�Ӷ��ھ����Ķ�������ϣ���Ŀ��8��
a��10-10)3cm3����������Znԭ�Ӷ��ھ����Ķ�������ϣ���Ŀ��8��![]() +6��
+6��![]() =4����Sԭ���ھ������ڲ�������4������һ����������4��ZnS��������Ϊ4��
=4����Sԭ���ھ������ڲ�������4������һ����������4��ZnS��������Ϊ4��![]() g�����ܶ�Ϊ
g�����ܶ�Ϊ g��cm-3��
g��cm-3��

����Ŀ������V��Ϊ����Ԫ�أ����γɶ��̬������ڹ�ҵ�����²��ϡ�����Դ�������й㷺Ӧ�á�
��1���������۵�ܸߣ��������ȷ�Ӧ�Ƶá�
��֪25����101 KPaʱ
4A1��s��+3O2��g���T2Al2O3��s����H1=akJmol-1
4V��s��+5O2��g���T2V2O5��s����H2=bkJmol-1
�������ȷ�Ӧұ������V��s�����Ȼ�ѧ����ʽΪ______��
��2��ȫ��Һ�������һ�����͵���ɫ��������ϵͳ������ԭ����ͼ��ʾ��
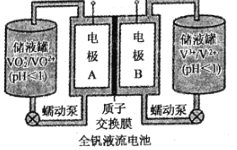
����������Ͽ�֪��
�������� | VO2+ | VO2+ | V3+ | V2+ |
��ɫ | ��ɫ | ��ɫ | ��ɫ | ��ɫ |
���õ�طŵ�ʱ��VO2+������ԭ��Ӧ���������ķ�Ӧʽ��______��
������ɴ���ʱ��������Һ����ɫΪ______��
����طŵ�ʱ����������Һ��pH��______������������������С����������������
���ø÷����������Ʒ�϶�ͭ������ƷӦ���ص�______��������A������B��������������ƿ�ʼʱ���缫������ȣ����һ��ʱ������缫����֮��Ϊ128g����ʱת�Ƶ��ӵ����ʵ���Ϊ______��