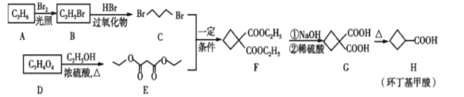
��Ŀ����
����Ŀ����������������Ҫ���л��ϳ��м��壬��ͼ����ϩ��A������D�ϳɻ����������·�ߡ�
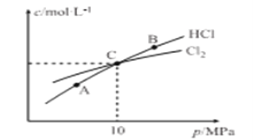
��ش��������⣺
(1)A�Ľṹ��ʽΪ____��
(2)B�й����ŵ�����Ϊ____��____��
(3)B��C�ķ�Ӧ����Ϊ____��D��E�ķ�Ӧ����Ϊ__________
(4)C+E��F�Ļ�ѧ����ʽΪ________
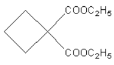
(5)��������������H������ͬ���칹��(�����������칹)����____�֣����к˴Ź���������3���Ľṹ��ʽΪ____��
����ʹ������Ȼ�̼��Һ��ɫ
������NaOH��Һ��Ӧ
������������Һ����������Ӧ
�ܲ�����״�ṹ
(6)�� ��
��![]() Ϊԭ�ϣ�ѡ�ñ�Ҫ�����Լ���д���ϳ�
Ϊԭ�ϣ�ѡ�ñ�Ҫ�����Լ���д���ϳ�![]() ��·��________(�ýṹ��ʽ��ʾ�л���ü�ͷ��ʾת����ϵ����ͷ��ע���Լ��ͷ�Ӧ����)
��·��________(�ýṹ��ʽ��ʾ�л���ü�ͷ��ʾת����ϵ����ͷ��ע���Լ��ͷ�Ӧ����)
���𰸡�CH3CH=CH2(��![]() ) ̼̼˫�� ��ԭ�� �ӳɷ�Ӧ ������Ӧ����ȡ����Ӧ�� BrCH2CH2CH2Br+
) ̼̼˫�� ��ԭ�� �ӳɷ�Ӧ ������Ӧ����ȡ����Ӧ�� BrCH2CH2CH2Br+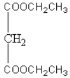
![]()
 +2HBr 8
+2HBr 8 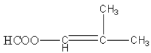
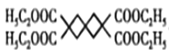
![]()
![]()
��������
A�ķ���ʽΪC3H6�������Ͷ�![]() ������̼̼˫��������ȡ����Ӧ����B��B�ٷ����ӳɷ�Ӧ�õ�C��D��C2H5OH����������Ӧ�õ�E��C��E��һ�������·���ȡ���õ�F�������õ�G��H���ɴ˽��з�����
������̼̼˫��������ȡ����Ӧ����B��B�ٷ����ӳɷ�Ӧ�õ�C��D��C2H5OH����������Ӧ�õ�E��C��E��һ�������·���ȡ���õ�F�������õ�G��H���ɴ˽��з�����
(1)����ͼʾת����ϵ��A��BΪȡ����Ӧ��B��CΪ�ӳɷ�Ӧ����A�ķ���ʽΪC3H6�������Ͷ�![]() ������̼̼˫������A�Ľṹ��ʽΪCH3CH=CH2(��
������̼̼˫������A�Ľṹ��ʽΪCH3CH=CH2(��![]() )���ʴ�Ϊ��CH3CH=CH2(��
)���ʴ�Ϊ��CH3CH=CH2(��![]() )��
)��
(2)B�Ľṹ��ʽΪBrCH2CH=CH2�����й�����Ϊ̼̼˫������ԭ�ӣ��ʴ�Ϊ��̼̼˫������ԭ�ӣ�
(3)B�����ӳɷ�Ӧ�õ�C��C��E��һ�������·���ȡ����Ӧ�õ�F���ʴ�Ϊ���ӳɷ�Ӧ��������Ӧ����ȡ����Ӧ����
(4) C��E��һ�������·���ȡ���õ�F����Ӧ����ʽΪ��BrCH2CH2CH2Br+
![]()
 +2HBr��
+2HBr��
(5)H�ķ���ʽΪC5H8O2������ʹ������Ȼ�̼��Һ��ɫ������̸̸�����ͼ������ݢ�����NaOH��Һ��Ӧ�������������Ȼ������ݢ�����������Һ����������Ӧ������ȩ�����������������������������������ܲ�����״�ṹ��֪����������ͬ���칹�庬��̼̼˫���ͼ����������ʸ�ͬ���칹��ɱ�ʾΪHCOOC4H7��������C4H7�к���һ��̼̼˫�������ڶ�ϩ��![]() ��CH3CH2CH=CH2��CH3CH=CHCH3���ֽṹ��ȥ��һ��H�õ��Ķ�ϩ����C4H7�ֱ���4�֡�2�֡�2�֣�����C4H7��8�֣�������������H��ͬ���칹�干�а��֣����к˴Ź���������3���Ľṹ��ʽΪ
��CH3CH2CH=CH2��CH3CH=CHCH3���ֽṹ��ȥ��һ��H�õ��Ķ�ϩ����C4H7�ֱ���4�֡�2�֡�2�֣�����C4H7��8�֣�������������H��ͬ���칹�干�а��֣����к˴Ź���������3���Ľṹ��ʽΪ ���ʴ�Ϊ��8��
���ʴ�Ϊ��8�� ��
��
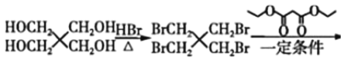
(6)��������ϩ��A������D�ϳɻ����������·�ߣ���������ɵó����� ��
��![]() Ϊԭ�Ϻϳ�
Ϊԭ�Ϻϳ�![]() ��·��Ϊ��
��·��Ϊ��

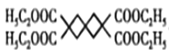
![]()
![]() ��
��

����Ŀ��������ѧ���������ϡ����ѧ������ȡ����Խ�ɾͣ�����Ϊ��ϡ�����Ԭ¡ƽ����ϡ��Ԫ�ذ����֡��ƺ���ϵԪ�ء���ش��������⣺
(1)д����̬����������(Sc2+)�ĺ�������Ų�ʽ____�����е���ռ�ݵĹ����Ϊ ____��
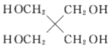
(2)�����������ⶨ��ϵԪ�غ�ʹ��ϵԪ�ط���ʱ������ʹ֮��ת���ɲ����Σ�Ȼ�����ն������������2LnCl3+3H2C2O4+nH2O=Ln2(C2O4)3nH2O+6HCl��
��H2C2O4��̼ԭ�ӵ��ӻ��������Ϊ____��1 mol H2C2O4�����к���������������Ŀ֮��Ϊ ___��
��H2O��VSEPRģ��Ϊ ___��д����H2O��Ϊ�ȵ������һ�������ӵĻ�ѧʽ_______��
��HCI��H2O�����γ�����ȶ���ˮ���������ξ��壬��HCl2H2O��HCl2H2O�к���H5O2+���ṹΪ![]() ���ڸ������У����ڵ���������___________
���ڸ������У����ڵ���������___________
a.��λ�� b.���Լ� c.�Ǽ��Լ� d.���Ӽ� e.������ f��� g.���»��� h.���� i.����
(3)�����г��˺˵����Ϊ21��25��Ԫ�ص���������ϼۣ�
Ԫ������ | �� | �� | �� | �� | �� |
Ԫ�ط��� | Sc | Ti | V | Cr | Mn |
�˵���� | 21 | 22 | 23 | 24 | 25 |
������� | +3 | +4 | +5 | +6 | +7 |
�Ա���������Ԫ��ԭ�ӵĺ�������Ų���Ԫ�ص���������ϼۣ��㷢�ֵĹ�����___________
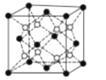
(4)PrO2(��������)�ľ����ṹ��CaF2���ƣ�������Pr(��)ԭ��λ�����ĺͶ��㡣������������Prԭ����Oԭ��֮��ľ���Ϊa pm����þ�����ܶ�Ϊ_____gcm-3(��NA��ʾ�����ӵ�������ֵ�����ؼ�������)��