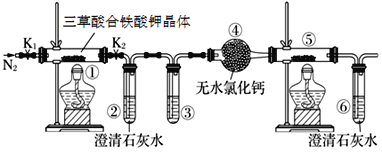
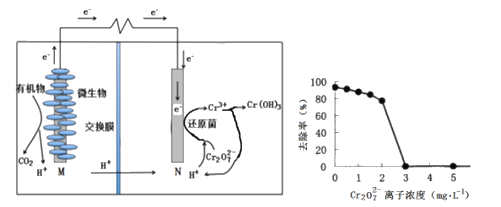
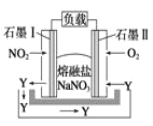
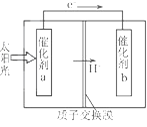
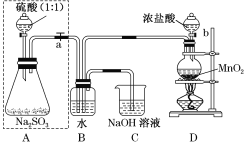
��Ŀ����
����Ŀ���Ѽ��仯�����ڻ�����ҽҩ�����ϵ��������Ź㷺��Ӧ�á�
��1����̬��ԭ�ӵļ۵����Ų�ʽΪ_____________������ͬ���ڵ�Ԫ���У���̬ԭ�ӵ�δ�ɶԵ�����������ͬ����____________�֡�
��2���ѱȸ��ᡢ����Ӳ����һ�����˵Ľṹ���ϣ��ѵ�Ӳ�ȱ������ԭ����_________��
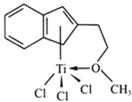
��3����Ũ��TiCl3��������Һ�м������ѣ���ͨ��HCl�����ͣ��ɵõ���λ��Ϊ6�����ΪTiCl3��6H2O����ɫ���壬�þ�����������������ʵ���֮��Ϊ1��5�����������ӵĻ�ѧʽΪ___________��
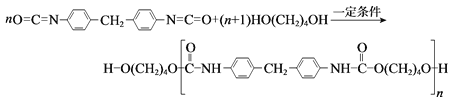
��4������Ľṹ����M�ܴ���ϩ����ϩ������ϩ�ľۺϣ���ṹ��ͼ��ʾ��
�����M��Ԫ���У��縺��������_________(������)��
��M��̼ԭ�ӵ��ӻ���ʽΪ____________��
��M���________(�����)��
a������ b������ c�����Ӽ� d����λ��
��5�����ʯ(TiO2)�Ǻ��ѵ���Ҫ����֮һ���侧���ṹ(��������ͬλ�õ�ԭ����ͬ)��ͼ��ʾ��
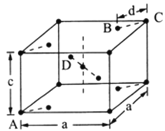
��A��B��C��D4������������ԭ����________(�����)��
����A��B��C��ԭ������ֱ�ΪA(0��0��0)��B(0.69a��0.69a��c)��C(a��a��c)����D��ԭ������ΪD(0.19a��____��___)���������ļ���d=______(�ô���ʽ��ʾ)��
���������ױ߳�Ϊa cm����Ϊc cm����TiO2������ܶ�Ϊ______g/cm3��
���𰸡�3d24s2 3 Tiԭ�ӵļ۵�������Al�࣬��������ǿ [TiCl(H2O)5]2+ �� sp2��sp3 c BD 0.81a 0.5c 0.31��![]() a
a ![]()
��������
(5)���ݾ����ṹ�������ķ������У���������ռ![]() ����������ռ
����������ռ![]() ���ڲ�����Ϊ�����������У����廯ѧʽΪTiO2��O����Ŀ��Ti�������������2����ԭ��λ�ھ����ڣ��������ϡ��µ����ϸ���2����ԭ�ӡ�
���ڲ�����Ϊ�����������У����廯ѧʽΪTiO2��O����Ŀ��Ti�������������2����ԭ��λ�ھ����ڣ��������ϡ��µ����ϸ���2����ԭ�ӡ�
(1)��Ϊ22��Ԫ�أ�λ�ڵ������ڣ���̬��ԭ�ӵļ۵����Ų�ʽΪ3d24s2����̬��ԭ��δ�ɶԵ���Ϊ3d�����2�����ӣ����������л�̬ԭ��δ�ɶԵ�����Ϊ2�Ļ��м۵����Ų�Ϊ3d84s2��Ni��3d104s24p2��Ge��3d104s24p4��Se��3�֣��ʴ�Ϊ��3d24s2��3��
(2)Tiԭ�ӵļ۵�������Al�࣬��������ǿ�������ѵ�Ӳ�ȱ����ʴ�Ϊ��Tiԭ�ӵļ۵�������Al�࣬��������ǿ��
(3)��λ��Ϊ6�����ΪTiCl36H2O����ɫ���壬�þ�����������������ʵ���֮��Ϊ1:5����������ֻ����3��Cl������������Ŀ�����H2O��H2O��Cl��ֵΪ5:1�����Ը������ӵĻ�ѧʽΪ[TiCl(H2O)5]2+���ʴ�Ϊ��[TiCl(H2O)5]2+��
(4)��M�к��е�Ԫ���У�Ti��Cl��O��C��H�����е縺��������O������Ϊ��Ԫ�أ��ʴ�Ϊ������
��M��C���ڵ�����˫����ǰ��Ϊsp3�ӻ�������Ϊsp2�ӻ����ʴ�Ϊ��sp2��sp3��
��M�к��й��ۼ�����λ�������۵���Ϊ����������˫����һ��Ϊ������һ��Ϊ����������M�в������Ӽ����ʴ�Ϊ��c��
(5)�ٸ��ݾ����ṹ�������ķ������У���������ռ![]() ����������ռ
����������ռ![]() ���ڲ�����Ϊ�����������У����廯ѧʽΪTiO2��O����Ŀ��Ti������������2����ԭ��λ�ھ����ڣ��������ϡ��µ����ϸ���2����ԭ�ӣ�����Ϊ2+4��
���ڲ�����Ϊ�����������У����廯ѧʽΪTiO2��O����Ŀ��Ti������������2����ԭ��λ�ھ����ڣ��������ϡ��µ����ϸ���2����ԭ�ӣ�����Ϊ2+4��![]() =4 ��Tiλ�����ĺͶ��㣬����Ϊ1+8��
=4 ��Tiλ�����ĺͶ��㣬����Ϊ1+8��![]() =2����A��B��C��D��������������ԭ����B��D���ʴ�Ϊ��BD��
=2����A��B��C��D��������������ԭ����B��D���ʴ�Ϊ��BD��
��A��B��C��ԭ������ֱ�ΪA(0��0��0)��B(0.69a��0.69a��c)��C(a��a��c)�����ʯ�����У�Ti�������ģ�����Ti��Χ��8��O�γɰ�����ṹ���ϵ���ṹΪ��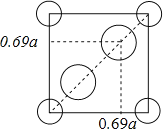 ���м���ĽṹΪ
���м���ĽṹΪ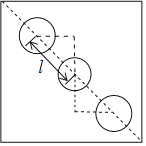 ��l��Ϊ�������ļ���d�������ϵ���ṹ��d=(a0.69a)��
��l��Ϊ�������ļ���d�������ϵ���ṹ��d=(a0.69a)��![]() =0.31��
=0.31��![]() a��D���ڸߵİ봦���ڵ�ƽ�棬�����м���Ľṹ������l=d=0.31��
a��D���ڸߵİ봦���ڵ�ƽ�棬�����м���Ľṹ������l=d=0.31��![]() a����֪D��x����Ϊ0.19a����y=a0.19a=0.81a������D������Ϊ(0.19a��0.81a��0.5c)���ʴ�Ϊ��0.81a��0.5c��0.31��
a����֪D��x����Ϊ0.19a����y=a0.19a=0.81a������D������Ϊ(0.19a��0.81a��0.5c)���ʴ�Ϊ��0.81a��0.5c��0.31��![]() a��
a��
���ɢٿ�֪��1�������к�2��Tiԭ�ӡ�4��Oԭ�ӣ���1������������=![]() g=
g=![]() g��1�����������=acm��acm��ccm=a2ccm3�������ܶ���=
g��1�����������=acm��acm��ccm=a2ccm3�������ܶ���=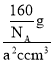 =
=![]() g��cm-3���ʴ�Ϊ��
g��cm-3���ʴ�Ϊ��![]() ��
��

 ����ʦ��Сһ����ʦ������ҵϵ�д�
����ʦ��Сһ����ʦ������ҵϵ�д� ���100�ֵ�Ԫ�Ż�������ϵ�д�
���100�ֵ�Ԫ�Ż�������ϵ�д�����Ŀ������ˮ�ܿ�(��Ҫ�ɷ�ΪCo2O3��������Fe2O3��Al2O3��MnO��MgO��CaO��SiO2��)������ȡ���ֻ����Լ�����ͼ�Dz����ܾ�����Ȼ��ܾ�����Ʊ����̣��ش��������⣺
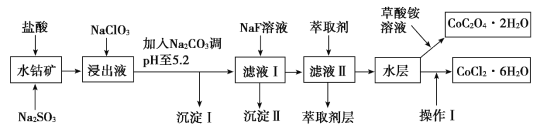
��֪���ٽ���Һ�к��е���������Ҫ��H+��Co2+��Fe2+��Mn2+��Al3+��Mg2+��Ca2+�ȡ�
�������в���������������������ʽ����ʱ��Һ��pH������
������ | Fe(OH)3 | Fe(OH)2 | Co(OH)2 | Al(OH)3 | Mn(OH)2 |
��ʼ���� | 2.7 | 7.6 | 7.6 | 4.0 | 7.7 |
��ȫ���� | 3.7 | 9.6 | 9.2 | 5.2 | 9.8 |
��1������������Co2O3������Ӧ�����ӷ���ʽΪ______________��
��2��1molNaClO3�ڽ���Һ�з�����Ӧʱ��ת�Ƶĵ�����Ϊ_________��
��3������Na2CO3��pH��5.2ֻ�õ����ֳ����������I�ijɷ�Ϊ______________���ѧʽ����ͬ������ȡ���㺬��Ԫ�أ������������Ҫ�ɷ�Ϊ______________��
��4����������������ˮ�����Ũ�������pHΪ2��3��________��________�����ˡ�ϴ�ӡ���ѹ��ɵȹ��̡�
��5����5.49g�����ܾ���(CoC2O4��2H2O)�������������ȣ����ȹ����У���ͬ�¶ȷ�Χ�ڵõ�һ�ֲ�ͬ�Ĺ������ʣ������������
�¶ȷ�Χ/�� | ��������/g |
150��210 | 4.41 |
290��320 | 2.41 |
���ⶨ���������ȹ��̲���������ֻ��ˮ������CO2����290��320����Χ�ڣ�ʣ��������ʵĻ�ѧʽΪ______________��