��Ŀ����
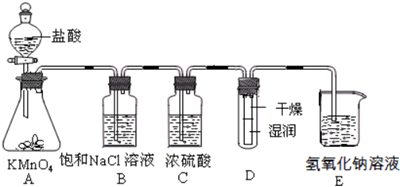
13����ҵ�Ͻ��������������ͨ�뵽NaOH��Һ�еõ�Ưˮ��ijͬѧ����ʵ����̽��Cl2���ʲ�ģ���Ʊ�Ưˮ����ͼ�Dz���ʵ��װ�ã���֪��2KMnO4+16HCl�T2MnCl2+2KCl+5Cl2��+8H2O
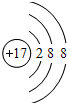
��1��Cl��±���еĴ���Ԫ�أ�д��������ӵĽṹʾ��ͼ
 ��
����2��װ��B��C�����÷ֱ��dz�ȥ�����е��Ȼ����������������
��3��д��װ��E�����Ļ�ѧ��Ӧ����ʽCl2+2NaOH=NaCl+NaClO+H2O��
��4��ʵ��ʱװ��D��ʪ��ĺ�ɫֽ����ɫ�����ﲿ��û����ɫ������һ��ʱ���ֽ��ȫ����ɫ����ͬѧ��ΪCl2���ܶȱȿ����������Թ��²�Cl2��Ũ�ȴ����Թ��ϲ�Cl2��Ũ�������µģ����жϸý����Ƿ��������������������ʵ�������ԭ��������ʵ�������Cl2��ʪ���ɫֽ���е�ˮ��ӦHClO����ʹʪ��ĺ�ɫֽ����ɫ�����ﲿ��û��HClO������ֽ������ɫ������һ��ʱ�䣬����ˮ�����˶��ᵼ������ֽ��ʪ�����ɫ
����Ľ����������Ʒ�����������ͨ��װ�и���ĺ�ɫֽ�����Թܣ���ͨ��װ��ʪ��ĺ�ɫֽ�����Թ��У�����Ϊ����������ʿɲ����𣩣�
���� ��1�������Ӻ�����17�����ӣ�������3�����Ӳ㣬����ֱ����ɵ���2��8��8���ݴ�д�������ӽṹʾ��ͼ��
��2��Ũ������лӷ��ԣ������ܹ��ٽ�Ũ����Ļӷ���������ȡ�������к����Ȼ����ˮ�������ñ���ʳ��ˮ��ȥ�Ȼ��⣬��Ũ��������ˮ��
��3���������������Ʒ�Ӧ�����Ȼ��ơ��������ƺ�ˮ��
��4��������������ʹ��ɫ������ɫ��HClOʹ��ɫ��������ɫ��ˮ���ӵ��˶���������
��� �⣺��1�������Ӻ�����17�����ӣ�������3�����Ӳ㣬����ֱ����ɵ���2��8��8���ṹʾ��ͼΪ�� ��
��
�ʴ�Ϊ�� ��
��
��2��Ũ������лӷ��ԣ������ܹ��ٽ�Ũ����Ļӷ���������ȡ�������к����Ȼ����ˮ������Ҫ�õ����﴿����������Ӧ����ͨ��ʢ�б���ʳ��ˮ��ϴ��ƿ��ȥ�Ȼ��⣬Ȼ��ͨ��ʢ��Ũ�����ϴ��ƿ��ȥˮ��
�ʴ�Ϊ����ȥ�����е��Ȼ������壻����������
��3���������������Ʒ�Ӧ�����Ȼ��ơ��������ƺ�ˮ����ѧ����ʽ��Cl2+2NaOH=NaCl+NaClO+H2O��
�ʴ�Ϊ��Cl2+2NaOH=NaCl+NaClO+H2O��
��4������������ʹ��ɫ������ɫ��HClOʹ��ɫ��������ɫ����һ��ʱ���ˮ�������˶�������ĺ�ɫֽ���У���ʵ�鲻������
�����ʵ�齫������ͨ��װ�и���ĺ�ɫֽ�����Թܣ���ͨ��װ��ʪ��ĺ�ɫֽ�����Թ��У�
�ʴ�Ϊ����������ʵ�������Cl2��ʪ���ɫֽ���е�ˮ��Ӧ����HClOʹʪ��ĺ�ɫֽ����ɫ�����ﲿ��û��HClO������ֽ������ɫ������һ��ʱ�䣬����ˮ�����˶��ᵼ������ֽ��ʪ�����ɫ��
��������ͨ��װ�и���ĺ�ɫֽ�����Թܣ���ͨ��װ��ʪ��ĺ�ɫֽ�����Թ��У�
���� ���⿼����������ȡʵ��װ�ü����������ʣ���ȷװ�õ����ü�װ���з����Ļ�ѧ��Ӧ���ɽ�𣬲��ؿ���ѧ������ʵ�����������Ŀ�ѶȲ���

��1����ҵ����ʱ����ȡ������һ����ӦΪ��CO+H2O��g��?CO2+H2��T��ʱ����1L�ܱ������г���0.2mol CO��0.3molˮ��������Ӧ����ƽ�����ϵ��c��H2��=0.12mol•L-1�����¶��´˷�Ӧ��ƽ�ⳣ��K=1�������������
��2�����᳧��β��ֱ���ŷŽ���Ⱦ������Ŀǰ��ѧ��̽������ȼ�������еļ���Ƚ��������ﻹԭΪ������ˮ���䷴Ӧ����Ϊ��
CH4��g��+4NO2��g���T4NO��g��+CO2��g��+2H2O��g����H=-574kJ•mol-1
CH4��g��+4NO��g���T2N2��g��+CO2��g��+2H2O��g����H=-1160kJ•mol-1
�����ֱ�ӽ�NO2��ԭΪN2���Ȼ�ѧ����ʽΪ��CH4��g��+2NO2��g��=CO2��g��+2H2O��g��+N2��g����H=-867kJ•mol-1��
��3�������ڴ�����ȼ�գ�����һ�ֵ��ʺ�ˮ����ѧ�����ô�ԭ������Ƴɰ���-����ȼ�ϵ�أ���ͨ�백���ĵ缫�ڼ��������·�����Ӧ�ĵ缫��ӦʽΪ2NH3+6OH--6e-=N2+6H2O��
�״���һ����Ҫ���л�����ԭ�ϣ���������ȡ���ѣ�һ���¶��£����������Ϊ1.0L�ĺ����ܱ������з�����Ӧ��2CH3OH��g��?CH3OCH3��g��+H2O��g����
| ���� ��� | �¶ȣ��棩 | ��ʼ���ʵ�����mol�� | ƽ�����ʵ�����mol�� | |
| CH3OH��g�� | CH3OCH3��g�� | H2O��g�� | ||
| �� | 387 | 0.20 | 0.080 | 0.080 |
| �� | 387 | 0.40 | a | b |
| �� | 207 | 0.20 | 0.090 | 0.090 |
��5��a=0.16
��6������˵������˵���÷�Ӧ�ﵽƽ��״̬����BD��������ĸ��
A��������ѹǿ���� B�����������c��CH3OCH3�� ����
C�����������ܶȲ��� D����λʱ��������CH3OH��CH3OCH3�ķ��Ӹ���֮��Ϊ2��1
��7����֪����������Ӧ��m��n������0����
��Ӧ�٣�CO��g��+2H2��g��?CH3OH��g����H=-m kJ•mol-1
��Ӧ�ڣ�2CO��g��+4H2��g��?CH3OCH3��g��+H2O��g����H=-n kJ•mol-1
������m��n�Ĺ�ϵ�У���ȷ����A������ĸ����
A��n��2m������ B��m��2n���� C.2m��n ���� D��m��3n��
| A�� | ��Һ�dzʵ����Եģ��������ǿ��Դ���� | |
| B�� | ���ˮ�м���FeCl3������Һ�������������Һ�ʺ��ɫʱ���õ�Fe��OH��3���� | |
| C�� | �峿����������Ҷ��ķ�϶���Բ��������ЧӦ��˵��������һ�ֽ��� | |
| D�� | ������������Һ�ͽ���ķ��������ö����ЧӦ�����ڻ�ѧ���� |
| A�� | ��״����6.72L NH3������ԼΪ5.1g | |
| B�� | 10g D216O����������������Ϊ4NA | |
| C�� | 2.3g������ȫ�����������ʧȥ�ĵ�����ĿΪ0.2NA | |
| D�� | 150mL1mol/L�Ȼ�����Һ��50mL1mol/L AlCl3��Һ�������ӵ����ʵ���Ũ����� |
| A�� | �Ȼ�����Һ�м��������ˮ��Al3++3NH3•H2O�TAl��OH��3��+3NH4+ | |
| B�� | ̼������ڴ���CaCO3+2H+�TCa2++CO2��+H2O | |
| C�� | �Ȼ�������Һ��ͨ��������Fe2++Cl2�TFe3++2Cl- | |
| D�� | �����ʯ��ˮ��ϡ���ᷴӦCa��OH��2+2H+�TCa2++2H2O |
| A�� | NaClˮ��Һ | B�� | ���� | C�� | �� | D�� | KOH���� |
��CO��g��+2H2��g���TCH3OH��g����H1
��CO2��g��+3H2��g���TCH3OH��g��+H2O��g����H2
��CO2��g��+H2��g���TCO��g��+H2O��g����H3
�ش��������⣺
| ��ѧ�� | H-H | C-O | C O | H-O | C-H |
| E/��kJ��mol-1�� | 436 | 343 | 1076 | 465 | 413 |
�ɴ˼����H1=___kJ��mol-1����֪��H2=-58kJ��mol-1�����H3=___kJ��mol-1��������
| A�� | 99��-41 | B�� | +99��+41 | C�� | -99��+41 | D�� | -99��41 |
