��Ŀ����
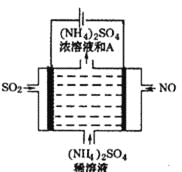
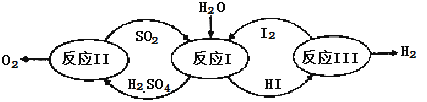
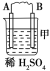
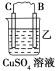
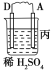
����Ŀ���Ʊ��屽��ʵ��װ������ͼ����Һ��Ӻ�ѹ��Һ©����������ʢ�б������۵���ƿA����Ӧ������A�е�Һ����к����������ɻ��������
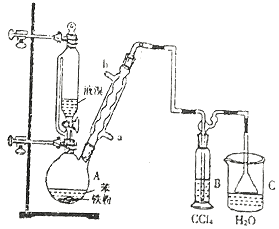
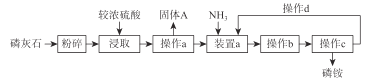
��1��д��A���Ʊ������Ļ�ѧ��Ӧ����ʽ______________��
��2�� B��ʢ�ŵ�CCl4��������______________
��3�������ܵ�������______________���������е�ˮӦ��______________�ڽ���(����a������b��)��
��4����C�м�����������Һ��֤������Һ����������ȡ����Ӧ�����Ǽӳɷ�Ӧ����Ӧ������______________��Ҳ��ʹ��______________��ֽ������֤���������ۡ�
��5���õ���������Ҫ�����²�����������������ˮϴ���ø����������10%NaOH��Һϴ����ȷ�IJ���˳����______________(����ĸ���)��
a.�٢ڢۢܢ� b.�ڢܢڢۢ� c.�ܢڢۢ٢� d.�ڢܢ٢ڢ�
���𰸡���1��![]() ��2����ȥ�廯�������е�������
��2����ȥ�廯�������е�������
��3������������a��4���е���ɫ�������ɣ���ɫʯ����ֽ����5��b
��������
�����������1������Һ���ڴ��������·�Ӧ�����屽����Ӧ����ѧ��Ӧ����ʽΪ![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��
��2����Ӧ�������б�����ӷ������� B��ʢ�ŵ�CCl4���Գ�ȥ�廯�������е����������ʴ�Ϊ����ȥ�廯�������е���������
��3���������ӷ����������������������������ã����ԭ�ϵ����������������е�ˮӦ��ѭ�½��ϳ����ʴ�Ϊ������������a��
��4����C�м�����������Һ�е���ɫ���廯���������ɣ�˵������Һ�巢������ȡ����Ӧ�����Ǽӳɷ�Ӧ���������ɵ��廯�⣬Ҳ��ʹ����ɫʯ����ֽ������ֽ��죬֤���������廯�⣬�ʴ�Ϊ���е���ɫ�������ɣ���ɫʯ����ֽ��
��5���õ����屽�л���������ʣ�������ˮϴ����ȥ�廯����������ˮ�����ʣ�����10%NaOH��Һϴ������ת��Ϊ������ˮ�����ʣ���ˮϴ��ȥ�������ƣ�����ø���������ȥˮ����ͨ��������뱽���屽����ѡb��

 ̽���빮�̺��Ͽ�ѧ����������ϵ�д�
̽���빮�̺��Ͽ�ѧ����������ϵ�д�