��Ŀ����
ij�����ӻ�������ɵĻ����ֻ���ܺ������������е������֣�K����NH4+��Mg2+��Ba2+��Cl����CO32����SO42����Ϊȷ������ɣ�ȷ��ȡ14.82g���������ˮ��300 mL������Һ���ֳ����ȷݷֱ��������ʵ�飺
��1����һ�ݼ���AgNO3��Һ�г�������
��2���ڶ��ݼ�����NaOH��Һ�����ȣ�����ʯ�Ҹ�����ռ������������1.12L
��3�������ݼ�����BaCl2��Һ��������ϴ�ӡ���������Ϊ6.27g����������м��������ᣬ�����ˡ�ϴ�ӡ���������Ϊ2.33 g
��������ʵ�飬�����Ʋⲻ��ȷ����
��1����һ�ݼ���AgNO3��Һ�г�������
��2���ڶ��ݼ�����NaOH��Һ�����ȣ�����ʯ�Ҹ�����ռ������������1.12L
��3�������ݼ�����BaCl2��Һ��������ϴ�ӡ���������Ϊ6.27g����������м��������ᣬ�����ˡ�ϴ�ӡ���������Ϊ2.33 g
��������ʵ�飬�����Ʋⲻ��ȷ����
| A����Һ��c(SO42��)Ϊ0.1mol/L��c(CO32��) Ϊ0.2mol/L |
| B���û�����в���Ba2+��Mg2+ |
| C��һ������NH4+�� K������ȷ��Cl���Ƿ���� |
| D��ʵ�飨3�������м��������ֻ���ˡ���ϴ�ӣ���Գ�NH4+����������Ӻ����IJⶨ���Ӱ�� |
A
�������������AgNO3��Һ�г���������������Cl-��CO32-��SO42-���ڼ�����NaOH��Һ���Ȳ������壬�����ǰ�������һ���������0.04mol���۲����������2.33gΪ���ᱵ�����ʵ�����0.01mol��6.27g���������ᱵ��̼�ᱵ��̼�ᱵ����Ϊ6.27g-2.33g=3.94g�����ʵ���Ϊ0.02mol����һ������CO32-��SO42-�����һ��û�� Mg2+��Ba2+��c(CO32-)=0.02��0.1=0.02��mol/L�����ٸ��ݵ���غ㣬�����Ϊ��n��+��=n��NH4+��=0.04mol��c��-��=2c(CO32-)+2x(SO42-)=0.06mol����һ����K+������0.02mol���ۺ����Ͽ��Եó���һ�����ڵ�������NH4+��K+��CO32-��SO42-��һ��û�е�����Mg2+��Ba2+�����ܴ���Cl-���ʱ���ѡ��Aѡ�

��ϰ��ϵ�д�
�����Ŀ

 Cr2O72��
Cr2O72��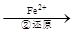 Cr3+
Cr3+ Cr(OH)3��
Cr(OH)3��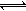 Cr2O72��(��ɫ)+H2O
Cr2O72��(��ɫ)+H2O