��Ŀ����
��51��2 g Cu��ȫ��������Ũ�����У��ռ�������������(��NO��N2O4��NO2)�Ļ���ﹲ0��8 mol����Щ����ǡ���ܱ�500 mL 2 mol/L NaOH��Һ��ȫ���գ������ķ�ӦΪ��2NO2��2NaOH=NaNO2��NaNO3��H2O��NO��NO2��2NaOH=2NaNO2��H2O��
�����ɵ�����Һ��NaNO2�����ʵ���Ϊ
| A��0��4 mol | B��0��6 mol |
| C��0��8 mol | D��0��2 mol |
C
����

 Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д� Сѧͬ�����������ܾ�ϵ�д�
Сѧͬ�����������ܾ�ϵ�д����й��ڼס��ҡ��������ĸ�ͼ���˵���У���ȷ����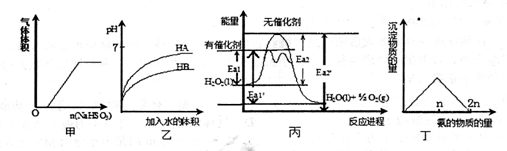
| A���������������Ʒ�ĩͶ��ϡ������Һ���й�ͼ�� |
| B�����ǵ�pH��HA��HB����������Һϡ��ʱpH�仯ͼ���ɴ˿�֪HA����������HB |
| C���ɱ���֪��˫��ˮ���д�������ʱ���Ȼ�ѧ����ʽΪ 2H2O2��1��="2H" 2O��l��+O2��g�� ��H=-2��Ea2����Ea2�� kJ��mol-l |
| D����������������Һ�еμӰ�ˮʱ�����������백�����ʵ����Ĺ�ϵͼ |
��Fe3+��Fe2+��NO3����NH4+��H+��H2O���������ֱ�����һ��������ԭ��Ӧ�еķ�Ӧ�����������������У�����ȷ����
| A����ԭ����ΪNH4+ |
| B���������뻹ԭ�������ʵ���֮��Ϊ1��8 |
| C������0.5mol NO3���μӻ�ԭ��Ӧ����ת�Ƶ���8mol |
| D�����Ѹ÷�Ӧ���Ϊ���أ���������ӦΪFe2+��e��Fe3+ |
���������Ҫ�ɷֵĻ�ѧʽΪFeO��Cr2O3��������SiO2��Al2O3�����ʡ���ҵ�ϳ����ù�������������ø�������Ʊ��ظ���أ�����Ϊ��
��֪����NaFeO2��ˮǿ��ˮ�⣻
���ظ����Ϊ�Ⱥ�ɫ��״���壬����ˮ���������Ҵ�����ǿ�����ԣ�
��2CrO42�� + 2H+ Cr2O72��+ H2O
Cr2O72��+ H2O
��1�����ղ���һ���������н��У����������ʺ�����������ʵ���������ϵ���
| A���� | B�������� | C��ʯӢ | D���մ� |
��д��Cr2O3�ʹ��������Ӧ����Na2CrO4�Ļ�ѧ����ʽΪ ��
��NaFeO2ˮ��ʱǿ��ˮ�����������������������ӷ���ʽΪ___ ___��
��3����Һ1�ijɷֳ�Na2CrO4��NaOH�⣬�����У��ѧʽ�� ��
��4��ͨ��;�����Na2CrO4��Һ���K2Cr2O7���壬���������У����ữ������KCl���������Ũ��������a�����ˡ�ϴ�ӡ����
�� ���ữ�������ô��������ҺpH��5����Ŀ���� ��
�ڲ���a������ ��
��֪��2Fe + 3Br2 = 2FeBr3��Fe2+�Ļ�ԭ�Դ���Br��������16.8 g����0.3 mol Br2��Ӧ�����ˮ�õ�������Һ��ͨ��a mol Cl2��������������ȷ����
| A����a = 0.1ʱ�������ķ�ӦΪ2Fe2++Cl2��2Fe3++2Cl�� |
| B����a = 0.45ʱ�������ķ�ӦΪ2Fe2++4Br��+3Cl2��2Fe3++2Br2+6Cl�� |
| C������Һ��Br����һ�뱻����ʱ�� c(Fe3+): c(Br��):c(Cl��) ��1:1:3 |
| D����0��a��0.15ʱ����Һ��ʼ������2c(Fe2+)+3c(Fe3+)+c(H+)��c(Cl��)+c(Br��)+ c(OH��) |
����Ϊ���ܻ�ԭ����NaBH4����ˮ����ˮ��Ӧ��NaBH4��2H2O=NaBO2��4H2��������˵������ȷ����(NaBH4��HΪ��1��)
| A��NaBH4�������������ǻ�ԭ�� |
| B��NaBH4����������H2O�ǻ�ԭ�� |
| C����Ԫ�ر���������Ԫ�ر���ԭ |
| D����������Ԫ���뱻��ԭ��Ԫ������֮��Ϊ1��1 |
��Ӧ�١��ڷֱ��ǴӺ���Һ�ij�ֿ�ʯ����ȡ�����Ҫ��Ӧ��
��2NaI��MnO2��3H2SO4=2NaHSO4��MnSO4��2H2O��I2
��2NaIO3��5NaHSO3=2Na2SO4��3NaHSO4��H2O��I2
����˵����ȷ����
| A��������Ӧ�о�Ϊ��Ԫ�ر����� |
| B����Ԫ���ڷ�Ӧ���б���ԭ���ڷ�Ӧ���б����� |
C�������ԣ�MnO2�� �� �� ��I2 ��I2 |
| D����Ӧ�١��������ɵ�����I2ʱת�Ƶ�������Ϊ1��5 |
��֪���з�Ӧ��Co2O3��6HCl(Ũ)=2CoCl2��Cl2����3H2O(��)��5Cl2��I2��6H2O=10HCl��2HIO3(��)������˵����ȷ���� (����)��
| A����Ӧ����HCl�������� |
| B����Ӧ����Cl2����������Ӧ |
| C����ԭ�ԣ�CoCl2��HCl��I2 |
| D�������ԣ�Co2O3��Cl2��HIO3 |
����������һ�ַ���������ɫ��������ʳƷ�й���ʱ����������Σ����������������
�»����NO��NO2�������й�˵���������(����)
| A��NaNO2�Ⱦ����������־��л�ԭ�� |
| B��ʳ�á����ﶹѿ�����ܻ�����������ƶ������Σ�� |
| C��NaNO2��θ�����õ����ӷ���ʽΪ�� 2NO2����2H��=NO����NO2����H2O |
| D��1 mol NaNO2��������������ȫ��Ӧ����NO��NO2��ת�Ƶ��ӵ����ʵ���Ϊ1 mol |