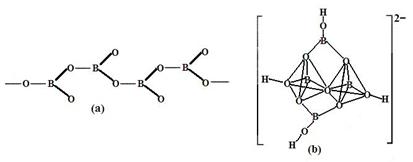
��Ŀ����
��13�֣���ͨ��״���£�X��Y��Z��������̬���ʡ�X�����Ԫ���ǵ�������ԭ�Ӱ뾶��С��Ԫ��(ϡ������Ԫ�س���)��Y��Z����Ԫ��R��ɣ���ӦY+2I-+2H+====I2+Z+H2O����ΪY�ļ�����Ӧ��W�Ƕ�����Ԫ�أ����������������ڲ���������������������ӶԵ�������X���ʵ����Ԫ��Ҫ����
��1�� Z�Ļ�ѧʽ__________________
��2����Y�Ͷ�������ֱ�ͨ��Ʒ����Һ������ʹƷ����ɫ����������ɫ����Һ����Y�Ͷ��������ʵ�鷽��:________________________________________________________��
��3���ٳ�ʵ��˵��X�������Ա�W����������ǿ(����һ����ѧ����ʽ��ʾ):_____________��
����ͼ��0.1 mol��L��1���ֵ������Һ��pH���¶ȱ仯��ͼ��
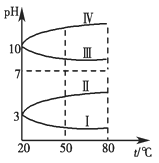
��1�����з���0.1 mol��L��1NH4Al(SO4)2��pH���¶ȱ仯��������________(��д���)��
��2��20 ��ʱ��0.1 mol��L��1NH4Al(SO4)2��2c(SO42-)��c(NH4+)��3c(Al3��)��________�������㾫ȷֵ��
��3������ʱ����100 mL 0.1 mol��L��1 NH4HSO4��Һ�еμ�0.1 mol��L��1 NaOH��Һ���õ���ҺpH��NaOH��Һ����Ĺ�ϵ������ͼ��ʾ��

�Է���ͼ��a��b��c��d�ĸ��㣬ˮ�ĵ���̶������________�㣻��b�㣬��Һ�и�����Ũ���ɴ�С������˳����______________________
��1�� Z�Ļ�ѧʽ__________________
��2����Y�Ͷ�������ֱ�ͨ��Ʒ����Һ������ʹƷ����ɫ����������ɫ����Һ����Y�Ͷ��������ʵ�鷽��:________________________________________________________��
��3���ٳ�ʵ��˵��X�������Ա�W����������ǿ(����һ����ѧ����ʽ��ʾ):_____________��
����ͼ��0.1 mol��L��1���ֵ������Һ��pH���¶ȱ仯��ͼ��

��1�����з���0.1 mol��L��1NH4Al(SO4)2��pH���¶ȱ仯��������________(��д���)��
��2��20 ��ʱ��0.1 mol��L��1NH4Al(SO4)2��2c(SO42-)��c(NH4+)��3c(Al3��)��________�������㾫ȷֵ��
��3������ʱ����100 mL 0.1 mol��L��1 NH4HSO4��Һ�еμ�0.1 mol��L��1 NaOH��Һ���õ���ҺpH��NaOH��Һ����Ĺ�ϵ������ͼ��ʾ��

�Է���ͼ��a��b��c��d�ĸ��㣬ˮ�ĵ���̶������________�㣻��b�㣬��Һ�и�����Ũ���ɴ�С������˳����______________________
��1��O2��2�֣�
��2��������ɫ�����Һ������Һ�ָ���ɫ����ԭͨ������ΪSO2������Һ����죬��ԭͨ��������O3��2�֣�
��3�� H2S+Cl2====S��2HCl��1�֣�(���������𰸾���)
��1����2�֣�
��2����10��3-10��11��mol��L��1 ��2�֣� ��3)a��2�֣���c(Na��)��c(SO42-)��c(NH4+)��c(OH��)��c(H��) ��2�֣�
��2��������ɫ�����Һ������Һ�ָ���ɫ����ԭͨ������ΪSO2������Һ����죬��ԭͨ��������O3��2�֣�
��3�� H2S+Cl2====S��2HCl��1�֣�(���������𰸾���)
��1����2�֣�
��2����10��3-10��11��mol��L��1 ��2�֣� ��3)a��2�֣���c(Na��)��c(SO42-)��c(NH4+)��c(OH��)��c(H��) ��2�֣�
���������I�����ȸ�����Ϣȷ��XΪCl2��RΪ��Ԫ�أ�YΪO3��ZΪO2��WΪ��Ԫ�ء�O3�����ǿ������ʹƷ����ɫ����SO2��ͬ���ʶ���ɫ�����Һ���м��ȣ�����Һ�ָ���ɫ����ԭͨ������ΪSO2������Һ����죬��ԭͨ��������O3����˵��Cl2�������Ա�S��������ǿ�ķ�Ӧ��H2S+Cl2====S��2HCl��K2S+Cl2====S��2KCl�ȡ�
���NH4Al(SO4)2��NH4+��Al3������ˮ��ʹ��Һ�����ԣ�pH��7�������¶����ߣ��ٽ�ˮ�⣬������ǿ��pH��С�����ߢ���ȷ��
�ƾ�NH4Al(SO4)2��Һ�еĵ���غ�ʽΪ2c(SO42-)+ c(OH��)��c(H+)+c(NH4+)+3c(Al3��)����2c(SO42-)��c(NH4+)��3c(Al3��)��c(H+) ��c(OH��)����1��10��3��1��10��11��mol��L��1��
����a��������ǡ�÷�Ӧ����(NH4)2SO4��NH4+����ˮ�⣬ˮ�ĵ���̶������b��[��a�㣨ǡ������(NH4)2SO4��֮�����Զ��һ��NaOH��ʹ����NH4+��OH����Ӧ����NH3��H2O����c(Na��)��c(SO42-)��c(NH4+)]���ָ�����ҺΪ���ԣ�����c(Na��)��c(SO42-)��c(NH4+)��c(OH��)��c(H��)��

��ϰ��ϵ�д�
 ��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�
��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�
�����Ŀ

 ���ж�HF
���ж�HF