��Ŀ����
T��ʱ���м�.�������ܱ������������������Ϊ1L�������������Ϊ2L���ֱ����.���������м���6mol A��3mol B��������Ӧ���£�
3A��g����b B��g��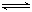 3C��g����2D��g����(bΪ
3C��g����2D��g����(bΪ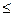 3��������) ��
3��������) ��
4min��������ڵķ�Ӧ�ﵽƽ�⣬A��Ũ��Ϊ2.4mol/L��B��Ũ��Ϊ1.8mol/L��
t min���������ڵķ�Ӧ�ﵽƽ�⣬B��Ũ��Ϊ0.8mol/L��
�����������Ϣ�ش��������⣺
��1���������з�Ӧ��ƽ������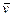 ��B���� ��
��B���� ��
��2�����¶�T��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��Ϊ__________________.
���±����ݿ�֪�÷�ӦΪ______________������ȡ������ȡ�����Ӧ��
��ѧƽ�ⳣ��K���¶�t�Ĺ�ϵ���±���
��3��T��ʱ������һ�����������ͬ�ı������У�Ϊ�˴ﵽƽ��ʱB��Ũ����ȻΪ0.8mol/L����ʼʱ����������м���C��D�����ʵ����ֱ�Ϊ3mol.2mol���������A.B�����ʵ����ֱ���___________.___________��
3A��g����b B��g��
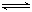 3C��g����2D��g����(bΪ
3C��g����2D��g����(bΪ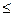 3��������) ��
3��������) ��4min��������ڵķ�Ӧ�ﵽƽ�⣬A��Ũ��Ϊ2.4mol/L��B��Ũ��Ϊ1.8mol/L��
t min���������ڵķ�Ӧ�ﵽƽ�⣬B��Ũ��Ϊ0.8mol/L��
�����������Ϣ�ش��������⣺
��1���������з�Ӧ��ƽ������
 ��B���� ��
��B���� ����2�����¶�T��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��Ϊ__________________.
���±����ݿ�֪�÷�ӦΪ______________������ȡ������ȡ�����Ӧ��
��ѧƽ�ⳣ��K���¶�t�Ĺ�ϵ���±���
| t�� | 700 | 800 | 830 | 1000 | 1200 |
| K | 0.6 | 0.9 | 1.0 | 1.7 | 2.6 |
��1��0.3mol/(L��min)�� ��2��10.8 ���� ��3��3mol.2mol
��

��ϰ��ϵ�д�
�����Ŀ
 C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc(A)="0.100" mol��L-1c(B)="0.200" mol��L-1 c(C)="0" mol��L-1����Ӧ��A��Ũ����ʱ��ı仯����ͼ��ʾ��
C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc(A)="0.100" mol��L-1c(B)="0.200" mol��L-1 c(C)="0" mol��L-1����Ӧ��A��Ũ����ʱ��ı仯����ͼ��ʾ��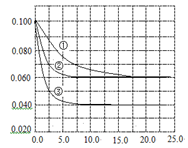
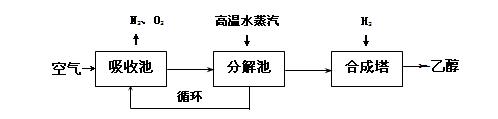
 CH3CH2OH(g)+H2O(g)
CH3CH2OH(g)+H2O(g)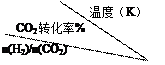
 Ur-(�����,aq) + H+(aq) ��37��ʱ��Ka =4.0��10-6��
Ur-(�����,aq) + H+(aq) ��37��ʱ��Ka =4.0��10-6��

 ʵ�����
ʵ����� ����3��ȡ1mL0.1mol/L KI��1mL0.1mol/LFeCl2��Һ���Թ��У���Ϻ����2~3��������ˮ����
����3��ȡ1mL0.1mol/L KI��1mL0.1mol/LFeCl2��Һ���Թ��У���Ϻ����2~3��������ˮ���� 2NO(g)+ O2(g)����˵���÷�Ӧ�Ѵﻯѧƽ��״̬����
2NO(g)+ O2(g)����˵���÷�Ӧ�Ѵﻯѧƽ��״̬���� 4NO(g)+6H2O(g)������������ȷ����
4NO(g)+6H2O(g)������������ȷ���� cC(g)+dD(g),C���ʵ�Ũ�ȣ�c%�����¶ȡ�ѹǿ�Ĺ�ϵ��ͼ��ʾ�������ж���ȷ���� �� ��
cC(g)+dD(g),C���ʵ�Ũ�ȣ�c%�����¶ȡ�ѹǿ�Ĺ�ϵ��ͼ��ʾ�������ж���ȷ���� �� ��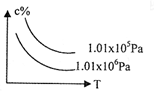
 2SO3��������2 mol SO3ʱ�ų�����ΪQ���ֽ�1 mol SO2��0.5 mol O2����ͬ�����·�Ӧ��ƽ��ų�������ΪQ1����
2SO3��������2 mol SO3ʱ�ų�����ΪQ���ֽ�1 mol SO2��0.5 mol O2����ͬ�����·�Ӧ��ƽ��ų�������ΪQ1����