��Ŀ����
��8�֣���ͼ������A��M��һ�������µ�ת����ϵ�����ֲ��P��Ӧ����δ�г��������У�I���ɵ�������Ԫ����ɵĵ������۵���ߵĽ����� K��һ�ֺ���ɫ���塣

����д���пհף�
��1�������ڱ��У���ɵ���G��Ԫ��λ�ڵ�__________����_________�塣
��2���ڷ�Ӧ�����������뻹ԭ�������ʵ���֮��Ϊ ��
��3���ڷ�Ӧ�ڡ��ۡ��ޡ����У������ڻ��Ϸ�Ӧ�����ڷ�������ԭ��Ӧ���� ������д��ţ�
��4����Ӧ�ܵ����ӷ���ʽ�ǣ� ��
��5����������D��KNO3��KOH���ڣ����Ƶ�һ�֡���ɫ��������Ч��ˮ��K2FeO4��������أ���ͬʱ������KNO2��H2O���÷�Ӧ�Ļ�ѧ����ʽ�ǣ� ��

����д���пհף�
��1�������ڱ��У���ɵ���G��Ԫ��λ�ڵ�__________����_________�塣
��2���ڷ�Ӧ�����������뻹ԭ�������ʵ���֮��Ϊ ��
��3���ڷ�Ӧ�ڡ��ۡ��ޡ����У������ڻ��Ϸ�Ӧ�����ڷ�������ԭ��Ӧ���� ������д��ţ�
��4����Ӧ�ܵ����ӷ���ʽ�ǣ� ��
��5����������D��KNO3��KOH���ڣ����Ƶ�һ�֡���ɫ��������Ч��ˮ��K2FeO4��������أ���ͬʱ������KNO2��H2O���÷�Ӧ�Ļ�ѧ����ʽ�ǣ� ��
��1���ģ���4������ ��2��1:2 ��3����
��4��3Fe2����NO3����4H�� = 3Fe3����NO����2H2O
��5��Fe2O3��3KNO3��4KOH =2K2FeO4��3KNO2��2H2O
��4��3Fe2����NO3����4H�� = 3Fe3����NO����2H2O
��5��Fe2O3��3KNO3��4KOH =2K2FeO4��3KNO2��2H2O
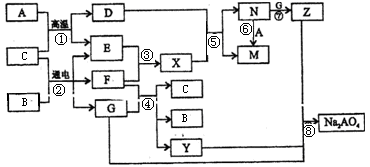
�������������������ͼ�⣬�ؼ�����ͻ�Ƶ㡣I���ɵ�������Ԫ����ɵĵ������۵���ߵĽ���������I��Al�� K��һ�ֺ���ɫ���壬��K��NO2�����Ը��ݷ�Ӧ���֪��J�����ᡣ���ݷ�Ӧ���֪��L��NO��������ʾ��֪��DӦ������������A��SO2�����ݷ�Ӧ�ڿ�֪��B����������������������ˮ�������ᣬ����C�����ᡣ��Ӧ�������ȷ�Ӧ������H����������G���������ݷ�Ӧ�ܿ�֪��MӦ������������������F�����Ρ���˸��ݷ�Ӧ�ݿ�֪��E���������������������ֽ⼴������������
��������ѧ�ƶ�����һ���ۺ��Խ�ǿ�����⣬��Ԫ�ؼ����������ʺ�������������������ѧ�����֪ʶ����������ѧ�Ƽ��ۺϡ��������ɿ���ѧ���Ի�ѧ֪ʶ������̶ȣ�����Ҫ��������ѧ�����ۺϷ���������˼ά���������ͼ��ķ�������ؼ�����Ѱ�ҡ�ͻ�ƿڡ�����ͻ�ƿڡ�����ץ���ء��֣�����������ɫ������״̬��������ζ�����ⷴӦ���������������Ʒ���������;�ȡ�

��ϰ��ϵ�д�
 Ӣ�żƻ���ĩ����ϵ�д�
Ӣ�żƻ���ĩ����ϵ�д�
�����Ŀ
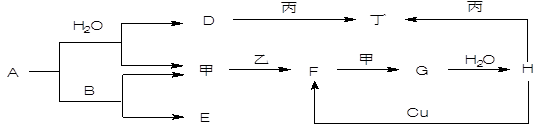
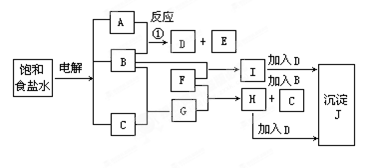
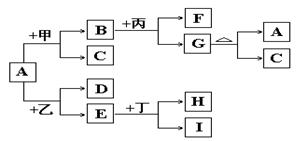
 A+C�Ļ�ѧ����ʽ�� ��
A+C�Ļ�ѧ����ʽ�� ��


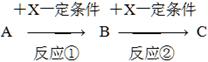
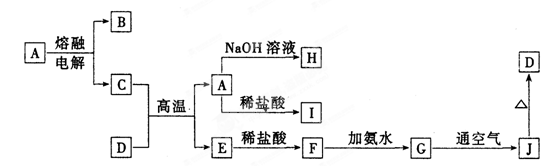 ��ش�
��ش�