��Ŀ����
����Ŀ��������ֲ��(�ر��Dzݱ�ֲ��)�����еijɷ֣����й㷺����;�����ᾧ��(H2C2O42H2O)��ɫ���۵�Ϊ101�棬������ˮ����������ˮ��������170�����Ϸֽ⡣���������ĵ��볣�� K1=5.4��10��2��K2=5.4��10��5���ش��������⣺
(1)��������װ�÷ֽ�����Ʊ�����������CO�������������˳��Ϊ__________(����ĸ���)��


![]()



(2)��ͬ�¶������£��ֱ���3֧�Թܰ�����Ҫ�����ʵ�飺
�Թ� | A | B | C |
�����Լ� | 4mL 0.01mol/L KMnO4 1ml 0.1moL/L H2SO4 2mL 0.1mol/L H2C2O4 | 4mL 0.02mol/L KMnO4 1ml 0.1moL/L H2SO4 2mL 0.1mol/L H2C2O4 | 4mL 0.03mol/L KMnO4 1ml 0.1moL/L H2SO4 2mL 0.1mol/L H2C2O4 |
��ɫʱ�� | 28�� | 30�� | ����ɫ |
д���Թ�B�з�����Ӧ�����ӷ���ʽ____________________������ʵ���ܷ�˵������ͬ�����£���Ӧ��Ũ��Խ��Ӧ����Խ���� __________(ѡ������������������)������������ɣ�_______________��
(3)���ʵ��֤������Ϊ����ķ��������������ȷ������___________��(�����)��
A�������£�ȡ0.010mol/L��H2C2O4��Һ������pH=2��
B�������£�ȡ0.010mol/L��NaHC2O4��Һ������pH >7��
C�������£�ȡpH=a(a<3)��H2C2O4��Һϡ��100������pH< a+2��
D������£�ȡ0.10mol/L��H2C2O4��Һ100mL������п�۷�Ӧ���ռ���H2���Ϊ224mL��
(4)Ϊ�ⶨijH2C2O4��Һ��Ũ�ȣ�ȡ20.00mL H2C2O4��Һ����ƿ�У�����2-3��ָʾ������0.1000mol/L��NaOH��Һ���еζ���������3��ƽ��ʵ�飬����NaOH��Һ����ֱ�Ϊ19.98mL��20.02mL��22.02mL��
������ָʾ��Ϊ__________���ζ��յ�ʱ������Ϊ____________________________________��
��H2C2O4��Һ���ʵ���Ũ��Ϊ__________��
�����в���������ⶨ���ƫ�ߵ���__________(�����)��
A���ζ�����ʢװNaOH��Һǰδ��ϴ
B���ζ������У���ƿ��̫���ң����²���Һ�彦��
C���ζ�ǰ������ȷ���ζ��յ�ʱ���Ӷ���
D���ζ�ǰ������ȷ���ζ��յ�ʱ���Ӷ���
���𰸡�B-E-D 2MnO4- +5H2C2O4 +6H+=2Mn2+ +10CO2��+8 H2O �� ʵ����KMnO4��Ũ��cB>cA�����䷴Ӧ������B>��A AC ��̪ ��ƿ����Һ����ɫ���(��)��ɫ���Ұ�����ڲ��仯 0.05000mol/L AD
��������
��1���������Ϣ��֪������ֽ�ʱ������ΪҺ̬�����ᾧ��ֽ����ɶ�����̼��һ����̼��ˮ��
��2��������Һ�и��������Һ�����������ɶ�����̼�����ᡢ����Ũ����ͬ���ı���������ҺŨ�ȷ�����Ӧ���ʱ仯�����������ҺŨ��Խ��Ӧ����Խ�죻
��3�����ݲ���Ϊ��Ԫ����Ͳ���������Һ�в�������������ˮ�������
��4����ǿ��ζ����ᵽ��Ӧ�յ����ɲ����ƣ����ɵ�Ϊǿ���������Լ��ԣ�
����H2C2O4��2NaOH������ϵʽ���ɵã�
�۵ζ���������������ʧ����Ϊ���ĵζ�������Һ����ı仯�����жϡ�
��1���������Ϣ��֪���������ȷֽ�ʱ�ۻ�ΪҺ̬����ѡ��װ��B���Ȳ��ᾧ�壻���ᾧ��ֽ����ɶ�����̼��һ����̼��ˮ��������ͨ��װ��E���ն�����̼��������ˮ���ռ�һ����̼�������������˳��ΪB-E-D���ʴ�Ϊ��B-E-D��
��2��������Һ�и��������Һ�����������ɶ�����̼����Ӧ�����ӷ���ʽΪ��2MnO4-+
5H2C2O4+6H+=2Mn2++10CO2��+8H2O���������ᡢ����Ũ����ͬ���ı���������ҺŨ�ȷ�����Ӧ���ʱ仯�����������ҺŨ��Խ��Ӧ����Խ�죬ʵ����KMnO4��Ũ��cB��cA�����䷴Ӧ���ʦ�B����A������ʵ����˵����ͬ�����£���Ӧ��Ũ��Խ��Ӧ����Խ�죬�ʴ�Ϊ��2MnO4-+5H2C2O4+6H+=2Mn2++10CO2��+8H2O���ܣ�ʵ����KMnO4��Ũ��cB��cA�����䷴Ӧ���ʦ�B����A��
��3��A������Ϊ��Ԫ�ᣬ��Ϊǿ������������Ũ��Ϊ0.02mol/L��pHС��2�������£�ȡ0.010mol/L��H2C2O4��Һ������pH=2��˵�����ڵ���ƽ�⣬֤����Ϊ���ᣬ��A��ȷ��
B�������£�0.010mol/L��NaHC2O4��Һ�в�������������ˮ�⣬��Һ�����ԣ� pHС��7����B����
C�������£�ȡpH=a��a��3����H2C2O4��Һϡ��100������pH��a+2��˵��ϡ�ʹٽ����룬��Һ�д��ڵ���ƽ�⣬Ϊ���ᣬ��C��ȷ��
D������£�ȡ0.10mol/L��H2C2O4��Һ100mL������п�۷�Ӧ��������ǿ�ỹ�����ᶼ�ռ���H2���Ϊ224mL����D����
��ѡAC���ʴ�Ϊ��AC��
��4����ǿ��ζ����ᵽ��Ӧ�յ����ɲ����ƣ����ɵ�Ϊǿ���������Լ��ԣ�����ѡ���̪��ָʾ�����������һ����ƿ����Һ����ɫ��ɣ��ۣ���ɫ���Ұ�����ڲ��仯���ʴ�Ϊ����̪����ƿ����Һ����ɫ��ɣ��ۣ���ɫ���Ұ�����ڲ��仯��
��ȡ20.00mLH2C2O4��Һ����ƿ�У�����2-3��ָʾ������0.1000mol/L��NaOH��Һ���еζ���������3��ƽ��ʵ�飬����NaOH��Һ����ֱ�Ϊ19.98mL��20.02mL��22.02mL������22.02mL���̫������ƽ�����Ϊ20ml����H2C2O4��2NaOH�ɵ�0.020L��c��2=0.1000mol/L��0.020L�����c=0.05000mol/L���ʴ�Ϊ��0.05000mol/L��
��A���ζ�����ʢװNaOH��Һǰδ��ϴ��������ҺŨ�ȼ�С�����ı���Һ������ⶨ���ƫ�ߣ�����ȷ��
B���ζ������У���ƿ��̫���ң����²���Һ�彦��������Һ��С�����ı���Һ�����С���ⶨ���ƫ�ͣ��ʴ���
C���ζ�ǰ������ȷ���ζ��յ�ʱ���Ӷ�������ȡ�ı���Һ�����С���ⶨ����Һ�Ѷ�ƫ�ͣ��ʴ���
D���ζ�ǰ������ȷ���ζ��յ�ʱ���Ӷ�������ȡ����Һ������ⶨ���ƫ�ߣ���D��ȷ��
��ѡAD���ʴ�Ϊ��AD

 ѧ�ڸ�ϰһ��ͨѧϰ�ܶ�Ա��ĩ������ӱ����������ϵ�д�
ѧ�ڸ�ϰһ��ͨѧϰ�ܶ�Ա��ĩ������ӱ����������ϵ�д� â���̸����������������ϵ�д�
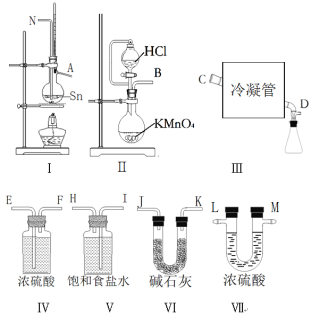
â���̸����������������ϵ�д�����Ŀ����ˮ���Ȼ���(SnCl4)�������л��ϳɵ��Ȼ�������ʵ���ҿ������ڵ�����Cl2��Ӧ�Ʊ�SnCl4��������ͼ�е������������װһ��ʵ��װ���Ʊ�SnCl4��ÿ��װ�����ʹ��һ�Σ���
��֪�����й������������±�
���� | ��ɫ��״̬ | �۵�/�� | �е�/�� | |
Sn | ����ɫ���� | 231.9 | 2260 | SnCl2��ˮ����SnCl4��ˮ�����ɹ�̬���������� ����Cl2��Ӧ���̷ų��������� |
SnCl4 | ��ɫҺ�� | ��33 | 114 | |
SnCl2 | ��ɫ���� | 246 | 652 |
��Fe3++Sn2+��Fe2++Sn4+ Fe2+ + Cr2O72- +H+��Fe3++Cr3++H2O��δ��ƽ��
�ش��������⣺
(1)������������������________��װ�â��з�����Ӧ�����ӷ���ʽΪ________��
(2)�ò��ܣ�δ��������������װ�ã���ȷ��˳���ǣ�����ӿڵĴ�����ĸ��_____��
(3)��μ���װ�õ�������______��ʵ�鿪ʼʱ�IJ���Ϊ_______��
(4)�������ȡ�����Ȼ���������¶�ڿ����У�Ԥ�ڿɿ����������dz��ְ�ɫ��������ѧ����ʽΪ_______��
(5)�����ظ���صζ����ⶨ��Ʒ�е�SnCl2�ĺ�����ȷ��ȡ����Ʒm g�����ձ��У�������Ũ�����ܽ⣬����������Ȼ�����Һ���ټ�ˮϡ�ͣ����Ƴ�250mL��Һ��ȡ25.00mL����ƿ�У���0.1000mol��L-1�ظ���ر���Һ�ζ����յ㣬���ı�Һ15.00mL�����Ʒ��SnCl2�ĺ���Ϊ____%(�ú�m�Ĵ���ʽ��ʾ)���ڲⶨ�����У��ⶨ�����ʱ���ӳ���С��ԭ����____�������ӷ���ʽ��ʾ����