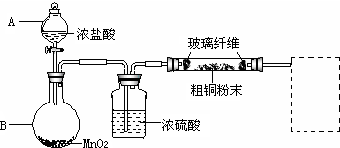
��Ŀ����
19�����ڻ����ķ�������ᴿ�������õķ����У����������ˡ���������ȡ�����ȷֽ⡢����ȣ����и�������ķ�����ᴿӦ����ʲô��������1��ʵ�����е�ʯ��ˮ���ã�Һ���ϳ�������CaCO3�������ù��˷�����ȥCa��OH��2��Һ��������CaCO3����
��2����ȡ��ˮ�еĵ⣬����ȡ������
��3����ȥʳ����Һ�е�ˮ���ɲ�������������
��4��������ˮ���ɲ���������
��5����ȥ�������е�̼��ƣ����ü��ȷֽⷽ����
��6����ȥNaCl�л��������Ĺ���⣬��������������
���� ��1��CaCO3������ˮ��������������ˮ��
��2�����������л��ܼ�����������ˮ��
��3��ˮ�ӷ���NaCl�ķе�ߣ�
��4��ʳ��ˮ������ķе�ͣ�
��5��̼��Ʋ��ȶ������ȿɷֽ����������ƣ�
��6������������
��� �⣺��1��CaCO3�Dz�����ˮ�����ʣ�Ҫ��ʯ��ˮ���룬���Բ��ù��˵ķ������ʴ�Ϊ�����ˣ�
��2��������ˮ���������ڱ������Ȼ�̼���л��ܼ����ұ������Ȼ�̼��ˮ�������ܣ�������ȡ�����룬�ʴ�Ϊ����ȡ��
��3����ȥʳ����Һ�е�ˮ�����������ķ������룬����ʱˮ�ӷ���ʣ���Ϊ�Ȼ��ƣ��ʴ�Ϊ��������
��4���Ӻ�ˮ�л�ȡ��ˮ����������ķ������룬����ʱˮ�ӷ�����ȴ��ɵõ�����ˮ���ʴ�Ϊ������
��5��̼��Ʋ��ȶ������ȿɷֽ����������ƣ����ü��ȵķ������룬�ʴ�Ϊ�����ȷֽ⣻
��6���������������ü��������ķ������룬�ʴ�Ϊ��������
���� ���⿼������ķ����ᴿ������ѡ��Ϊ��Ƶ���㣬�������ʵ����ʡ����ʲ���Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬ע�ⳣ�����뷽��������������Ӧ�ã���Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ֥�鿪���γ�������ϵ�д�
֥�鿪���γ�������ϵ�д� ����ѧ��ţ��Ӣ��ϵ�д�
����ѧ��ţ��Ӣ��ϵ�д�
�����Ŀ
8����pH=1����Һ�У����Դ���������������ǣ�������
| A�� | Na+��K+��S2-��Cl- | B�� | K+��Na+��SO42-��NO3- | ||
| C�� | K+��Na+��SiO32-��NO3- | D�� | Ba2+��Mg2+��SO42-��Cl- |
7���ܱ������з��Ϸ�Ӧ��2A��g��+B��g��?xC��g�������2molA��1molB�ﵽƽ���C���������Ϊw%����ά���������ݻ����¶Ȳ��䣬����ʼ���ʵ���A��0.6mol��B��0.3mol��C��1.4mol�����������ﵽƽ���C�����������Ϊw%����x��ֵΪ��������
| A�� | 2��4 | B�� | 3��4 | C�� | 2��3 | D�� | ��ȷ�� |
14���������ӷ���ʽ��ȷ���ǣ�������
| A�� | ϡ�������þƬ�ϣ�Mg+2H+�TMg2++H2�� | |
| B�� | ����þ��Һ������������Һ��Ӧ��Ba2++SO42-�TBaSO4�� | |
| C�� | ��������Һ�����ᷴӦ��Ag++Cl-�TAgCl�� | |
| D�� | ����ʯ��ˮ��̼������Һ��Ӧ��Ca��OH��2+CO32-�TCaCO3��+H2O |
4��������ijͬѧ��NaHSO4���������ͽ��еķ��������в���ȷ���ǣ�������
| A�� | ����Ԫ����ɿ��ж��ǻ����� | |
| B�� | ��������������������ַ����ε���ɣ��ʿ��������� | |
| C�� | �����������������������ַ����ε���ɣ��ʿ����������� | |
| D�� | NaHSO4����ˮ�ɵ���������ӹʿ��Գ�Ϊ�� |
11�����й���±�ص�������ȷ���ǣ�������
| A�� | ±���е��ʴ�������Ȼ���� | |
| B�� | ��˵�������ӣ������۷е����� | |
| C�� | ��˵�������ӣ�������������ǿ | |
| D�� | ������ˮ��Ӧ��������ͨʽX2+H2O�THX+HXO��ʾ |
8����ӦC��s��+H2O��g��?CO��g��+H2��g����һ�ݻ��ɱ���ܱ������н��У��������������������£����������ĸı���䷴Ӧ���ʼ�����Ӱ����ǣ�������
| A�� | ����������䣬����H2O��g�������ʵ��� | |
| B�� | �������������Сһ�� | |
| C�� | ����������䣬����N2ʹ��ϵѹǿ���� | |
| D�� | ����ѹǿ���䣬����N2ʹ���������� |
9�����и����е����ӣ�������Һ�д���������ǣ�������
| A�� | H+��Ca2+��Cl-��CO32- | B�� | Na+��Fe3+��OH-��SO42- | ||
| C�� | K+��Na+��OH-��Cl- | D�� | Cu2+��Ba2+��Cl-��SO42- |