��Ŀ����
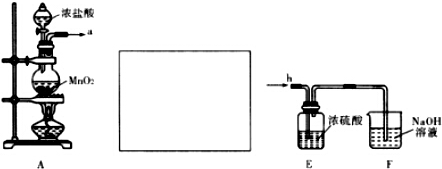
15�� ij��ѧС���������������������װ�ã���ͼ�����Ի������Ʊ�����ϩ
ij��ѧС���������������������װ�ã���ͼ�����Ի������Ʊ�����ϩ��֪��
| �ܶȣ�g/cm3�� | �۵㣨�棩 | �е㣨�棩 | �ܽ��� | |
| ������ | 0.96 | 25 | 161 | ������ˮ |
| ����ϩ | 0.81 | -103 | 83 | ������ˮ |
��12.5mL�����������Թ�A�У��ټ���l mLŨ���ᣬҡ�Ⱥ�������Ƭ��
������������Ӧ��ȫ�����Թ�C�ڵõ�����ϩ��Ʒ��
��A�����Ƭ�������Ƿ�ֹ���У�����B���˵�������е�������������
���Թ�C���ڱ�ˮԡ�е�Ŀ����ʹ����ϩҺ�������ٻӷ���
��2���Ʊ���Ʒ
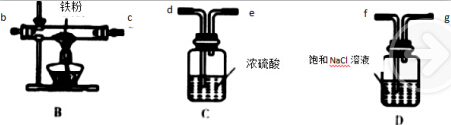
�ٻ���ϩ��Ʒ�к��л������������������ʵȣ����뱥��ʳ��ˮ������ �á��ֲ㣬����ϩ���ϲ㣬��Һ����c �������ţ�ϴ�ӣ�
a��KMnO4��Һ b��ϡH2SO4 c��Na2CO3��Һ
��ʵ���ƵõĻ���ϩ��Ʒ�����������������۲���������ܵ�ԭ����c
a������ʱ��100�濪ʼ�ռ���Ʒ
b��������ʵ����������
c���Ʊ���Ʒʱ���������Ʒһ������
��3���������ֻ���ϩ��Ʒ�ʹ�Ʒ�ķ�������������bc��a�������Ը��������Һ b���ý����� c���ⶨ�е㣮
���� ��1���ٸ�������ϩʵ���֪ʶ������װ��A�����Ƭ�������Ƿ�ֹ���У��������ɵĻ���ϩ�ķе�Ϊ83�棬Ҫ�õ�Һ̬����ϩ������B���˵���������������ã����ڻ���ϩ������
�ڱ�ˮԡ��Ŀ���ǽ��ͻ���ϩ�������¶ȣ�ʹ��Һ����
��2�������ڷ�Һ��ϩ��Ʒ�л�������������ͻ��������ᴿ����ʱ��c��Na2CO3��Һ��ϴ�ӿɳ�ȥ�
��a������ǰ�ռ�����Ʒ�л������ʣ�ʵ�ʲ����������۲�����
b����ȡ�Ļ���ϩ���ʵ�������ʵ���ƵõĻ���ϩ��Ʒ�����������۲�����
c���ֲ�Ʒ�л��л����������²ⶨ���ĵĻ������������ƵõĻ���ϩ��Ʒ�����������۲�����
��3�����ݻ����û�й̶��ķе㣬���������й̶��ķе㣬�ݴ˿��жϲ�Ʒ�Ĵ��ȣ�
��� �⣺��1���ٸ�������ϩʵ���֪ʶ������װ��A�����Ƭ�������Ƿ�ֹ���У��������ɵĻ���ϩ�ķе�Ϊ83�棬Ҫ�õ�Һ̬����ϩ������B���˵���������������ã����ڻ���ϩ������
�ʴ�Ϊ����ֹ���У�������
�ڱ�ˮԡ��Ŀ���ǽ��ͻ���ϩ�������¶ȣ�ʹ��Һ�����ʴ�Ϊ��ʹ����ϩҺ�������ٻӷ���
��2�������ڷ�Һ��ϩ��Ʒ�л�������������ͻ����������룺�Ʊ����������ᴿ����ʱ��c��Na2CO3��Һ��ϴ�ӿɳ�ȥ�ᣬ�ʴ�Ϊ��c��
��a������ʱ��70�濪ʼ�ռ���Ʒ����ǰ�ռ�����Ʒ�л������ʣ�ʵ�ʲ����������۲�������a����
b��������ʵ���������ˣ���ȡ�Ļ���ϩ�����ʵ�������ʵ���ƵõĻ���ϩ��Ʒ�����������۲�������b����
c�����ֲ�Ʒ�л��л����������²ⶨ���ĵĻ������������ƵõĻ���ϩ��Ʒ�����������۲�������c��ȷ��
�ʴ�Ϊ��c��
��3�������Ʒ�뾫Ʒ�ɼ�������ƣ��۲��Ƿ�������������������壬���Ǿ�Ʒ��������ݻ����û�й̶��ķе㣬���������й̶��ķе㣬ͨ���ⶨ����ϩ��Ʒ�ͻ���ϩ��Ʒ�ķе㣬Ҳ���жϲ�Ʒ�Ĵ��ȣ�
�ʴ�Ϊ��bc��
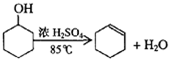
���� ���⿼�����Ի������Ʊ�����ϩ��ʵ�鷽�����ۺϿ��������ʵķ��뷽���������������ķ����ȣ��Ѷ����У�����ѧ�����ʵ�������������

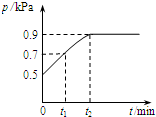 ��1.0L�ܱ������з���0.1mol X����һ���¶��·�����Ӧ��X��g��?Y��g��+Z��g����H��0 ������������ѹǿp�淴Ӧʱ��t�ı仯��ϵ��ͼ��ʾ�����·�����ȷ���ǣ�������
��1.0L�ܱ������з���0.1mol X����һ���¶��·�����Ӧ��X��g��?Y��g��+Z��g����H��0 ������������ѹǿp�淴Ӧʱ��t�ı仯��ϵ��ͼ��ʾ�����·�����ȷ���ǣ�������| A�� | t1ʱn��X��=0.04 mol | |
| B�� | t1��t2����������ƽ����Է��������� | |
| C�� | �����ƽ����ϵ��Y�ĺ�������������ϵ�¶Ȼ����Z���� | |
| D�� | �����������䣬�ٳ���0.1 mol ����X��ƽ�������ƶ���X��ת���ʼ��� |
| A�� | ��ΪSiO2��H2SiO3����������SiO2����ˮ��H2SiO3 | |
| B�� | CO2ͨ��ˮ������Na2SiO3��ˮ��Һ���п��Եõ�������� | |
| C�� | ��Ϊ����ʱ������Ӧ��Na2CO3+SiO2$\frac{\underline{\;����\;}}{\;}$Na2SiO3+CO2�������Թ�������Ա�̼��ǿ | |
| D�� | SiO2���������������������� |
| A�� | 0.2mol•L-1Na2SO4��Һ�к���Na+��SO42-�����ʵ���Ϊ0.6mol | |
| B�� | 50mL2mol•L-1NaCl��Һ��100mL0.5mol•L-1MgCl2��Һ�У�Cl-�����ʵ���Ũ����� | |
| C�� | ��1Lˮ����22.4L�Ȼ������������Ũ����1mol•L-1 | |
| D�� | 10g98%�����ᣨ�ܶ�Ϊ1.84g•cm-3����10mL18.4mol•L-1�������Ũ������ͬ�� |
| A�� | 6mol | B�� | 10mol | C�� | 3amol | D�� | 3mol |
| A�� | ��ʯ�� | B�� | �ɱ� | C�� | ʯ��ʯ | D�� | Ư�� |
| A�� | ������ѹǿ����ʱ��仯���仯 | |
| B�� | ��λʱ������0.1mol Xͬʱ����0.3mol Z | |
| C�� | ������X��Y��Z��Ũ��֮��Ϊ1��2��3 | |
| D�� | �����ڸ����ʵ�Ũ�Ȳ���ʱ��仯���仯 |

 ��
��