��Ŀ����
��ҵ�Ͽ������÷����е�CO2Ϊԭ����ȡ�״����䷴Ӧ����ʽΪ��CO2+3H2 CH3OH+H2O����ش��������⣺
CH3OH+H2O����ش��������⣺
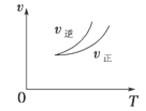
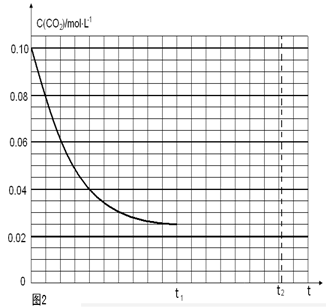
��1����֪���³�ѹ�����з�Ӧ�������仯����ͼ��ʾ��
 д���ɶ�����̼�������Ʊ��״����Ȼ�ѧ����ʽ__ _��
д���ɶ�����̼�������Ʊ��״����Ȼ�ѧ����ʽ__ _��
�÷�Ӧ�ġ�S____0(�>����<����=��)���� ______����������ڸ÷�Ӧ�Է����С�
��2�����������Ӧ����ʽ��ƽ�ⳣ��Kֵ�����÷�Ӧ__ ��ѡ���ţ���
A��һ��������Ӧ�����ƶ� B����ƽ���ƶ�ʱ����Ӧ������������С
C��һ�����淴Ӧ�����ƶ� D����ƽ���ƶ�ʱ�淴Ӧ�����ȼ�С������
��3�����������Ӧ�����������ܱ������з�������˵����Ӧ�Ѵﵽƽ��״̬���� __ ��ѡ���ţ���
A��3v��(H2)=v��(CO2) B��C(H2) = C(CO2)
C��������������ܶȲ��� D��������ѹǿ����
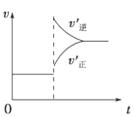
��4������Ӧ�������ݻ�Ϊ2.0L����Ӧʱ��4.0 min��������������ܶ�������2.0g/L�������ʱ����CO2��ƽ����Ӧ����Ϊ ����Ӧ��t1ʱ�ﵽƽ�⣬������c(CO2)��ʱ��t�仯����������ͼ��ʾ�����������������䣬t1ʱ���������ѹ����1L���뻭��t1��c(CO2)��ʱ��t�仯��������(t2�ﵽ�µ�ƽ��)��
 CH3OH+H2O����ش��������⣺
CH3OH+H2O����ش��������⣺��1����֪���³�ѹ�����з�Ӧ�������仯����ͼ��ʾ��
|
|
|
|

 д���ɶ�����̼�������Ʊ��״����Ȼ�ѧ����ʽ__ _��
д���ɶ�����̼�������Ʊ��״����Ȼ�ѧ����ʽ__ _���÷�Ӧ�ġ�S____0(�>����<����=��)���� ______����������ڸ÷�Ӧ�Է����С�
��2�����������Ӧ����ʽ��ƽ�ⳣ��Kֵ�����÷�Ӧ__ ��ѡ���ţ���
A��һ��������Ӧ�����ƶ� B����ƽ���ƶ�ʱ����Ӧ������������С
C��һ�����淴Ӧ�����ƶ� D����ƽ���ƶ�ʱ�淴Ӧ�����ȼ�С������
��3�����������Ӧ�����������ܱ������з�������˵����Ӧ�Ѵﵽƽ��״̬���� __ ��ѡ���ţ���
A��3v��(H2)=v��(CO2) B��C(H2) = C(CO2)
C��������������ܶȲ��� D��������ѹǿ����
��4������Ӧ�������ݻ�Ϊ2.0L����Ӧʱ��4.0 min��������������ܶ�������2.0g/L�������ʱ����CO2��ƽ����Ӧ����Ϊ ����Ӧ��t1ʱ�ﵽƽ�⣬������c(CO2)��ʱ��t�仯����������ͼ��ʾ�����������������䣬t1ʱ���������ѹ����1L���뻭��t1��c(CO2)��ʱ��t�仯��������(t2�ﵽ�µ�ƽ��)��

��1��3H2(g) + CO2(g)= CH3OH(l)+ H2O(l)����H=��50KJ/mol��<�� ���£���2��A��D��
��3��CD����4��0.01mol.L-1.min-1����ͼ
��3��CD����4��0.01mol.L-1.min-1����ͼ
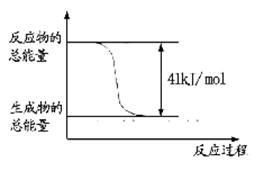
�����������1����������ɵ��Ȼ�ѧ����ʽ����CO(g)+H2O(l)=CO2(g)+H2(g) ��H=��41KJ/mol; ��CO(g) +H2(g) =CH3OH(l)��H=��91KJ/mol; �ڣ��٣������ɵ�3H2(g) + CO2(g)= CH3OH(l)+ H2O(l)����H=��50KJ/mol;�ɷ���ʽ��֪���÷�Ӧ��һ����ϵ�Ļ��ҳ̶ȼ�С�ķ�Ӧ�����ԡ�S<0�����ڸ÷�Ӧ������Ӧ�Ƿ��ȷ�Ӧ�����Է�Ӧ�ڵ�������������ڸ÷�Ӧ�Է����С���2�����ڸ÷�Ӧ������Ӧ�Ƿ��ȷ�Ӧ������������Ӧ����ʽ��ƽ�ⳣ��Kֵ�����ƽ�������ƶ�������Kֻ���¶��йأ�����ѹǿ��Ũ�ȵ��أ�����ֻ���¶Ƚ��Ͳſ�������������������ʱ��V����V�涼��С��V���С�Ķ࣬V��>V�棬ƽ�������ƶ����淴Ӧ�����ȼ�С�����������ӡ����ѡ��ΪA��D����3��A������Ӧ�ﵽƽ�⣬��v��(H2)= 3v��(CO2)������B�����ڶ�������ʱ�ǰ���3:1�����ʵ����Ĺ�ϵ���ĵģ������ڿ�ʼ���������������ֻ�а���ijһȷ���ı�����ϣ��ﵽƽ��ʱ���й�ϵ��C(H2) = C(CO2)����˲�����Ϊ�ж�ƽ��ı�־������C��������������Һ̬���ʣ�����Ӧδ�ﵽƽ�⣬������������ͻᷢ���仯��������ܶ�Ҳ�ᷢ���ı䡣���������������ܶȲ��䣬������Ϊ�ж�ƽ��ı�־����ȷ��D���÷�Ӧ�Ƿ�Ӧǰ������������ȵķ�Ӧ�����δ�ﵽƽ�⣬����������ʵ����ͻᷢ���仯���������������ѹǿ�ͻ�ı䡣���������ѹǿ���������Ϊ�ж�ƽ��ı�־����ȷ����4����Ӧ�������ݻ�Ϊ2.0L��������������ܶ�������2.0g/L�������������������2.0g/L��2.0L=4.0g.�������ӵ�CO2������Ϊ
 �����Ԧ�n(CO2)=" ��m��M="
�����Ԧ�n(CO2)=" ��m��M=" 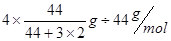 =0.08mol,���V(CO2)= ��c(CO2)�¦�t="(0.08mol" ��2L)��4min = 0.01mol/(L��min)��
=0.08mol,���V(CO2)= ��c(CO2)�¦�t="(0.08mol" ��2L)��4min = 0.01mol/(L��min)����ͼ���֪����ӦCO2(g)+3H2(g)
 CH3OH(l)+H2O(l)����ʼʱc(CO2)=0.10mol/L��ƽ��ʱc(CO2)=0.025mol/L����c(CO2)=0.075mol/L������ƽ��ʱ�����ʵ�Ũ��Ϊ��c(H2)=0.075mol/L���÷�Ӧ��ƽ�ⳣ��
CH3OH(l)+H2O(l)����ʼʱc(CO2)=0.10mol/L��ƽ��ʱc(CO2)=0.025mol/L����c(CO2)=0.075mol/L������ƽ��ʱ�����ʵ�Ũ��Ϊ��c(H2)=0.075mol/L���÷�Ӧ��ƽ�ⳣ��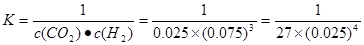 �������������������䣬t1ʱ���������ѹ����1L��c(CO2)=0.05mol/L��c(H2)=0.15mol/L��ƽ�������ƶ���������c(CO2)=xmol/L,������c(H2)=3xmol/L��ƽ��ʱ�����ʵ�Ũ�ȷֱ�Ϊc(CO2)="(0.05-x)mol/L" ��c(H2)="(0.15-3x)mol/L" ��ƽ�ⳣ�����䡣��
�������������������䣬t1ʱ���������ѹ����1L��c(CO2)=0.05mol/L��c(H2)=0.15mol/L��ƽ�������ƶ���������c(CO2)=xmol/L,������c(H2)=3xmol/L��ƽ��ʱ�����ʵ�Ũ�ȷֱ�Ϊc(CO2)="(0.05-x)mol/L" ��c(H2)="(0.15-3x)mol/L" ��ƽ�ⳣ�����䡣�� �����x= 0.025mol/L����t2�ﵽ�µ�ƽ��ʱc(CO2)=0.025mol/L.��t1��c(CO2)��ʱ��t�仯��������Ϊ��ͼ��ʾ��
�����x= 0.025mol/L����t2�ﵽ�µ�ƽ��ʱc(CO2)=0.025mol/L.��t1��c(CO2)��ʱ��t�仯��������Ϊ��ͼ��ʾ�� 2Ũ����ʱ��ͼ��ı�ʾ��
2Ũ����ʱ��ͼ��ı�ʾ��
��ϰ��ϵ�д�
�����Ŀ
 CH3OH(g)+H2O(g)
CH3OH(g)+H2O(g)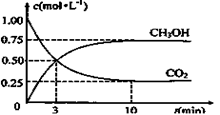
 2G(g)���й��������£�
2G(g)���й��������£� CH3OCH3(g)��H2O(g)
CH3OCH3(g)��H2O(g)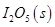 �����ڼ��CO����Ӧԭ��Ϊ��
�����ڼ��CO����Ӧԭ��Ϊ��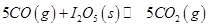
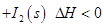 ����2L�ܱ������м�������
����2L�ܱ������м�������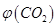 ��ʱ��ı仯����ͼ��ʾ��
��ʱ��ı仯����ͼ��ʾ��
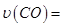 _____________��
_____________�� ���������
���������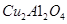 Ϊ���������Խ�
Ϊ���������Խ�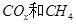 �Ļ������ֱ��ת��Ϊ���ᡣ
�Ļ������ֱ��ת��Ϊ���ᡣ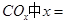 ______________��
______________�� ________14(�>������<����=��)�����¶��´���ĵ��볣��K=__________(�ú�a��b��ʽ�ӱ�ʾ)��
________14(�>������<����=��)�����¶��´���ĵ��볣��K=__________(�ú�a��b��ʽ�ӱ�ʾ)��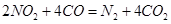 ���Դ�������β���������÷�Ӧ���Ϊԭ��أ�������Na2O������ʣ��������缫��ӦʽΪ________________________________��
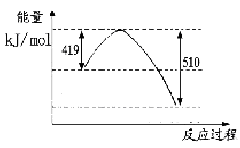
���Դ�������β���������÷�Ӧ���Ϊԭ��أ�������Na2O������ʣ��������缫��ӦʽΪ________________________________�� 2SO3(g) ��H����196kJ/mol����߷�Ӧ��SO2��ת���ʣ��Ǽ���SO2�ŷŵ���Ч��ʩ��
2SO3(g) ��H����196kJ/mol����߷�Ӧ��SO2��ת���ʣ��Ǽ���SO2�ŷŵ���Ч��ʩ��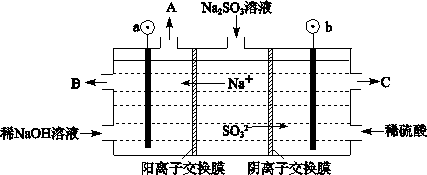
 2Z(g)������˵����ȷ���� (����)
2Z(g)������˵����ȷ���� (����)