̀âÄ¿ÄÚÈƯ
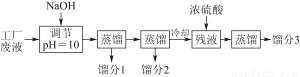
ʵÑéỂÀûÓĂẠ́Ë᳧ÉƠÔü£¨Ö÷̉ª³É·ÖΪ̀úµÄÑơ»¯Îï¼°ÉÙÁ¿FeS¡¢SiO2µÈ£©ÖƱ¸¾Û̀ú£¨¼îʽẠ́Ëá̀úµÄ¾ÛºÏÎºÍẦ·¯£¨FeSO4¡¤7H2O£©£¬¹ư³̀ÈçÏ£º

£¨1£©½«¹ư³̀¢ÚÖĐ²úÉúµÄÆø̀åͨÈëÏÂÁĐÈÜ̉ºÖĐ£¬ÈÜ̉º»áÍÊÉ«µÄÊÇ________¡£
A£®Æ·º́ÈÜ̉º B£®×ÏɫʯÈïÈÜ̉º
C£®ËáĐÔKMnO4ÈÜ̉º D£®äåË®
£¨2£©¹ư³̀¢ÙÖĐ£¬FeSºÍO2¡¢H2SO4·´Ó¦µÄ»¯Ñ§·½³̀ʽΪ_____________________________¡£
£¨3£©¹ư³̀¢ÛÖĐ£¬Đè¼ÓÈëµÄÎïÖÊÊÇ________¡£
£¨4£©¹ư³̀¢ÜÖĐ£¬Ơô·¢½á¾§Đè̉ªÊ¹ÓĂ¾Æ¾«µÆ¡¢Èư½Å¼Ü¡¢ÄàÈư½Ç£¬»¹Đè̉ªµÄ̉ÇÆ÷ÓĐ________________¡£
£¨5£©¹ư³̀¢Ưµ÷½ÚpH¿ÉÑ¡ÓĂÏÂÁĐÊÔ¼ÁÖеÄ________£῭îÑ¡ÏîĐ̣ºÅ£©¡£
A£®Ï¡Ạ́Ëá B£®CaCO3 C£®NaOHÈÜ̉º
£¨6£©¹ư³̀¢̃ÖĐ£¬½«ÈÜ̉ºZ¼ÓÈȵ½70¡«80 ¡æ£¬Ä¿µÄÊÇ______________________¡£
£¨7£©ÊµÑéỂΪ²âÁ¿ËùµĂµ½µÄ¾Û̀úÑùÆ·ÖĐ̀úÔªËصÄÖÊÁ¿·ÖÊư£¬½øĐĐÏÂÁĐʵÑé¡£
¢ÙÓĂ·ÖÎö̀́ƽ³ÆÈ¡2£®700 gÑùÆ·£»
¢Ú½«ÑùÆ·ÈÜÓÚ×ăÁ¿µÄÑÎËáºó£¬¼ÓÈë¹ưÁ¿µÄÂÈ»¯±µÈÜ̉º£»
£¨1£©ACD
£¨2£©4FeS£«3O2£«6H2SO4=2Fe2£¨SO4£©3£«6H2O£«4S
£¨3£©Fe£¨»̣̀ú£©¡¡£¨4£©Ơô·¢Ăó¡¢²£Á§°ô¡¡£¨5£©B
£¨6£©´Ù½øFe3£«µÄË®½â
£¨7£©31£®1%
¡¾½âÎö¡¿£¨1£©¹ư³̀¢ÚÖĐ²úÉúµÄÆø̀åÊÇSO2£¬¾ßÓĐƯ°×ĐÔ£¨ÄÜʹƷº́ÈÜ̉ºÍÊÉ«£©£»SO2¶ÔËá¼îָʾ¼ÁĂ»ÓĐƯ°××÷ÓĂ¡£
£¨2£©FeS»¯ºÏ¼ÛÉư¸ßΪ1£«2£½3, O2 »¯ºÏ¼Û½µµÍ4£¬Á½Ơ߸öÊư±ÈÊÇ4¡Ă3£¬ÔÙ½áºÏÔ×ÓÊغăÅäƽ¡£
£¨3£©¹ư³̀¢ÛÖĐ¼ÓÈë̀ú½«Fe3£«»¹ÔΪFe2£«£¬ÓÖ²»̉ưÈëÔÓÖÊ¡£
£¨4£©Ơô·¢ÈÜ̉ºÊ±½«̉º̀å·ÅÔÚƠô·¢ĂóÖĐ£¬²¢²»¶ÏÓĂ²£Á§°ô½Á°è¡£
£¨5£©ÖĐºÍ¹ưÁ¿Ạ́ËᣬѡÔñCaCO3¹̀̀壬ÈôÑ¡ÔñNaOH ÈÜ̉º£¬Ộ̉×ʹFe3£«³Áµí¡£
£¨6£©Éư¸ßζȣ¬´Ù½øFe3£«µÄË®½â¡£
£¨7£©ÓÉ̉ÑÖª¿ÉµĂm£¨BaSO4£©£½3£®495 g£¬Ộn£¨SO42-£©£½0£®015 mol£¬¸ù¾Ư×é³ÉÖªn£¨Fe£©£½n£¨SO42-£©£½0£®015 mol£¬m£¨Fe£©£½0£®84 g£¬̀úÔªËصÄÖÊÁ¿·ÖÊưΪ0£®84 g¡Â2£®700 g¡Á100%£½31£®1%¡£

 ̀́̀́Ị̈ÉÏ̉»±¾ºĂ¾íϵÁĐ´đ°¸
̀́̀́Ị̈ÉÏ̉»±¾ºĂ¾íϵÁĐ´đ°¸ Đ¡Ñ§Éú10·ÖÖÓÓ¦ÓẰâϵÁĐ´đ°¸
Đ¡Ñ§Éú10·ÖÖÓÓ¦ÓẰâϵÁĐ´đ°¸Ä³Ñ§Ï°Đ¡×éÓûÄ£Äâ´Óij¹¤³§·Ï̉ºÖĐ»ØÊƠ±ûͪ¡¢̉̉´¼ºÍ̉̉ËáµÄʵÑé¡£Öƶ¨ÁËÈçÏÂÊÔÑéÁ÷³̀¡£
̉ÑÖª¸Ă·Ï̉ºÖĐÖ÷̉ªº¬ÓĐ̉̉´¼£¬ÆäÖĐ»¹ÈÜÓбûͪ¡¢̉̉ËáºÍ̉̉Ëá̉̉ơ¥¡£Ç̉¸÷ÖÖ³É·ÖµÄ·ĐµăÈçÏÂ±í£º
ÎïÖÊ | ±ûͪ | ̉̉Ëá̉̉ơ¥ | ̉̉´¼ | ̉̉Ëá |
·Đµă£¨¡æ£© | 56.2 | 77.06 | 78 | 117.9 |
£¨1£©Áó·Ö3µÄ³É·ÖΪ____________¡£
£¨2£©ÉÏÊöÁ÷³̀Öе÷½ÚpH£½10µÄÄ¿µÄÊÇ________________________________________________________________________________________________________________________________________________¡£
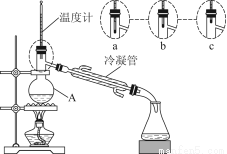
£¨3£©¸ĂĐ¡×éͬѧµÄƠôÁó×°ÖĂÈçͼËùʾ¡£ỘAÖĐζȼƵÄλÖĂƠưÈ·µÄÊÇ________£῭î¡°a¡±¡°b¡±»̣¡°c¡±£©¡£
£¨4£©¹ú¼̉±ê×¼¹æ¶¨£¬ÓÅÖʸ߶ÈŨÏăĐÍ°×¾Æ×ÜËáÁ¿£¨̉Ổ̉Ëá¼Æ£©Ó¦²»ÉÙÓÚ0.30 g/L£¬×Üơ¥Á¿£¨̉Ổ̉Ëá̉̉ơ¥¼Æ£©Ó¦²»ÉÙÓÚ2.0 g/L¡£
¢ÙΪ²â¶¨Ä³°×¾ÆÑùÆ·µÄ×ÜËáÁ¿£¬È¡20.00 mLÑùÆ·ÓÚ׶ĐÎÆ¿ÖĐ£¬¼ÓÈë·Ó̀ªÖ¸Ê¾¼Á2µÎ£¬ÓĂ0.010 mol/LµÄNaOH±ê×¼ÈÜ̉ºµÎ¶¨ÖÁÖƠµă¡£ÅжÏÖƠµăµÄ̉À¾ƯÊÇ________________________________________________________________________________________________________________________________________________¡£
Èô¸Ă°×¾ÆÑùƷΪÓÅÖʼ¶£¬ỘÏûºÄNaOHÈÜ̉º̀å»ưÓ¦²»Đ¡ÓÚ________mL¡£
¢Ú°×¾ÆÖеÄ×Üơ¥Á¿¿ÉÓĂ·µµÎ·¨²â¶¨¡£ÍùÉÏ̀âµÎ¶¨ºóµÄÈÜ̉º£¨Ç¡ºĂÖÁÖƠµă£©ÖĐÔÙ¼ÓÈë20.00mL0.100mol/L NaOH±ê×¼ÈÜ̉º£¬ÓĂͼװÖĂˮԡ¼ÓÈÈ°ëĐ¡Ê±¡£ÀäÈ´ºóÓĂ0.100mol/LµÄẠ́Ëá±ê×¼ÈÜ̉ºµÎ¶¨ÖÁÖƠµă¡£¼ÓÈÈ°ëĐ¡Ê±µÄÄ¿µÄÊÇ______________________£¬ÀäÄư¹ÜµÄ×÷ÓĂÊÇ______________¡£̉ÑÖª×îÖƠÏûºÄẠ́Ëá±ê×¼ÈÜ̉º7.70 mL£¬¸Ă°×¾ÆÑùÆ·ÖĐ×Üơ¥Á¿Îª________g/L£¨±£ÁôĐ¡ÊưµăºóÈưλÊư×Ö£©¡£
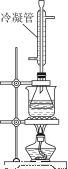
£¨5£©ÏÂÁĐ²Ù×÷»áʹ×Üơ¥Á¿²â¶¨½á¹ûÆ«¸ßµÄÊÇ________£¨Ñ¡̀î±àºÅ£©
a£®¼ÓÈÈʱδʹÓĂˮԡºÍÀäÄư¹Ü
b£®µÎ¶¨Ç°µÎ¶¨¹ÜÄÚÎ̃ÆøÅƯ£¬µÎ¶¨ºó²úÉúÆøÅƯ
c£®µÎ¶¨¹ÜδÓĂẠ́Ëá±ê×¼ÈÜ̉ºÈóÏ´