��Ŀ����
�����14�֣�
���ж��ֺ����ᣬ�����ᣨH2SO3�������ᣨH2SO4���������ᣨH2SO4��SO3����������ᣨH2S2O3���ȵȣ�����������Ϊ��Ҫ���ڹ�ҵ���й㷺��Ӧ�á���ʵ���ң�Ũ�����dz��õĸ������
������м��㣺
(1)�����ᣨH2SO4��SO3������ˮ�����е�SO3��ת��Ϊ���ᡣ����445g����������ˮ���4.00L���ᣬ����������ʵ���Ũ��Ϊ________mol/��
(2)����Ũ������ˮ�����ɵ�H2SO4��H2O���㣬250g��������Ϊ98%�����������ն���gˮ��
(3)�������ǹ�ҵ�����������Ҫԭ�ϡ��������������յĻ�ѧ��Ӧ���£�
3FeS2��8O2��Fe3O4��6SO2 4FeS2��11 O2��2Fe2O3��8SO2
��48mol FeS2��ȫ��Ӧ��������2934.4L����״���������㷴Ӧ������Fe3O4��Fe2O3���ʵ���֮�ȡ�
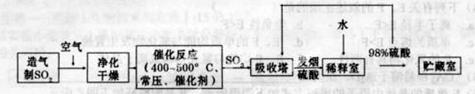
(4)��������ȡ���ᣬ���ܳ��������Դ���ܱ�����������һ�ֺ��з�չǰ;���Ʊ�����ķ�����
�����������Ϊ0.84�Ļ�����壨H2S��H2O��N2���ڿ�������ȫȼ�գ�����������77%���������������SO2���������ˮ�����壩������֪������ɣ�N2�������0.79��O2�������0.21��
���ж��ֺ����ᣬ�����ᣨH2SO3�������ᣨH2SO4���������ᣨH2SO4��SO3����������ᣨH2S2O3���ȵȣ�����������Ϊ��Ҫ���ڹ�ҵ���й㷺��Ӧ�á���ʵ���ң�Ũ�����dz��õĸ������
������м��㣺
(1)�����ᣨH2SO4��SO3������ˮ�����е�SO3��ת��Ϊ���ᡣ����445g����������ˮ���4.00L���ᣬ����������ʵ���Ũ��Ϊ________mol/��
(2)����Ũ������ˮ�����ɵ�H2SO4��H2O���㣬250g��������Ϊ98%�����������ն���gˮ��
(3)�������ǹ�ҵ�����������Ҫԭ�ϡ��������������յĻ�ѧ��Ӧ���£�
3FeS2��8O2��Fe3O4��6SO2 4FeS2��11 O2��2Fe2O3��8SO2
��48mol FeS2��ȫ��Ӧ��������2934.4L����״���������㷴Ӧ������Fe3O4��Fe2O3���ʵ���֮�ȡ�
(4)��������ȡ���ᣬ���ܳ��������Դ���ܱ�����������һ�ֺ��з�չǰ;���Ʊ�����ķ�����
�����������Ϊ0.84�Ļ�����壨H2S��H2O��N2���ڿ�������ȫȼ�գ�����������77%���������������SO2���������ˮ�����壩������֪������ɣ�N2�������0.79��O2�������0.21��
(1)1.25 (2)250��98%��98��16��40g
(3)2934.4��22.4��131mol
��Fe3O4 amol Fe2O3 bmol
3a+2b��48 8a+11��2b��131 a��4 b��18
n (Fe3O4):n(Fe2O3)��2:9
(4)������Ϊ1�����0.84���������ȫȼ������0.84������������0.84���ˮ������1.26����������������Ϊ1.26��0.21��1.77��10.62
��� ��0.84�£�10.62��1.26+1.84����0.075
��0.84�£�10.62��1.26+1.84����0.075
(3)2934.4��22.4��131mol
��Fe3O4 amol Fe2O3 bmol
3a+2b��48 8a+11��2b��131 a��4 b��18
n (Fe3O4):n(Fe2O3)��2:9
(4)������Ϊ1�����0.84���������ȫȼ������0.84������������0.84���ˮ������1.26����������������Ϊ1.26��0.21��1.77��10.62
���
 ��0.84�£�10.62��1.26+1.84����0.075
��0.84�£�10.62��1.26+1.84����0.075���������(1)445g����������ʵ�����445g��178g/mol��2.5mol����������������������2.5mol��2.5mol���������ֲ���2.5mol���ᣬ����Һ����������ʵ�����5.0mol��Ũ����5.0mol��4L��1.25mol/L��
(2)250g��������Ϊ98%�������������������250g��98%��245g�������ܼ�ˮ��5g����������ʵ�����245g��98g/mol��2.5mol�����Խ��ˮ�����ʵ�����2.5mol��������2.5mol��18g/mol��45g����˻���������ˮ��������45g��5g��40g��
(3)��Fe3O4�����ʵ�����amol��Fe2O3�����ʵ�����bmol
��Ӧ���������������ʵ�����2934.4L��22.4L/mol��131mol
����ݷ���ʽ��֪3a+2b��48��8a+11��2b��131
���a��4��b��18
��n (Fe3O4):n(Fe2O3)��2:9
(4)������Ϊ1����������Ȼ�����ȫȼ�յķ���ʽ2H2S��3O2��ȼ2SO2��2H2O ��֪��0.84���������ȫȼ������0.84������������0.84���ˮ������1.26�����������Ӧ�п�������77%������������Ϊ
 ��1.77��10.62������������������
��1.77��10.62������������������ ��
�� ��0.075
��0.075
��ϰ��ϵ�д�
�����Ŀ
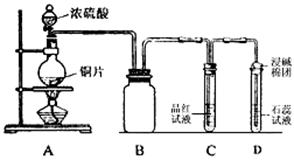

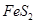 ��ȫȼ�շų�7.2kJ������
��ȫȼ�շų�7.2kJ������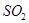 �������Ϊ7%��O2Ϊ11%��N2Ϊ82%����
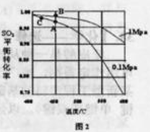
�������Ϊ7%��O2Ϊ11%��N2Ϊ82%����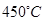 ���ң���Ӧͼ��A�㣩����û��ѡ��
���ң���Ӧͼ��A�㣩����û��ѡ��