��Ŀ����
��ijͬѧΪ̽��ͭ��Ũ����ķ�Ӧ���������ͼ��ʾװ�ý������й�ʵ��
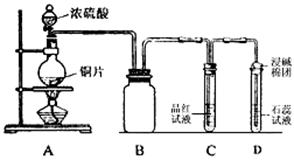
��1��B�������ռ�ʵ�������ɵ������װ�ã���δ�����ܻ�ȫ�����ڴ�����ϰѵ��ܲ���
���� ��
��2����д��ͭ��Ũ���ᷴӦ�Ļ�ѧ����ʽ ��
��3��ʵ����D�е����� ��
��4��ʵ���У���ͬѧȡ6.4gͭƬ��12mL 18mol��L-1H2SO4��Һ����Բ����ƿ�й��ȣ�ֱ����Ӧֹͣ���������ƿ�л���ͭƬʣ�ࣻ��ͬѧ������ѧ�Ļ�ѧ֪ʶ�жϻ�����һ����H2SO4��ʣ�࣬����ҩƷ���ܹ�������֤��Ӧֹͣ�����ƿ��ȷ��ʣ���H2SO4���� ����ĸ��ţ���
a��BaCl2��Һ b��Ba(NO3)2��Һ c������ d��Na2CO3��ĩ
��5������16gͭ��50mLH2SO4���ʵ���Ũ��Ϊһ��ֵ��Ũ���ᷴӦ��ͭ��ȫ�ܽ⡣��ش�
�ٷ�Ӧ�в����������ڱ�״���µ����Ϊ L��
�ڸ÷�Ӧ�б���ԭ��H2SO4�����ʵ���Ϊ mol��
�۴�����������ȫ���ͷź�����Һ�еμ�VmL a mol��L-1NaOH��Һ��ǡ��ʹ��Һ��
��Cu2+ȫ��ת��Ϊ��������ԭŨ������H2SO4�����ʵ���Ũ��= mol��L-1��

��1��B�������ռ�ʵ�������ɵ������װ�ã���δ�����ܻ�ȫ�����ڴ�����ϰѵ��ܲ���
���� ��
��2����д��ͭ��Ũ���ᷴӦ�Ļ�ѧ����ʽ ��
��3��ʵ����D�е����� ��
��4��ʵ���У���ͬѧȡ6.4gͭƬ��12mL 18mol��L-1H2SO4��Һ����Բ����ƿ�й��ȣ�ֱ����Ӧֹͣ���������ƿ�л���ͭƬʣ�ࣻ��ͬѧ������ѧ�Ļ�ѧ֪ʶ�жϻ�����һ����H2SO4��ʣ�࣬����ҩƷ���ܹ�������֤��Ӧֹͣ�����ƿ��ȷ��ʣ���H2SO4���� ����ĸ��ţ���
a��BaCl2��Һ b��Ba(NO3)2��Һ c������ d��Na2CO3��ĩ
��5������16gͭ��50mLH2SO4���ʵ���Ũ��Ϊһ��ֵ��Ũ���ᷴӦ��ͭ��ȫ�ܽ⡣��ش�
�ٷ�Ӧ�в����������ڱ�״���µ����Ϊ L��
�ڸ÷�Ӧ�б���ԭ��H2SO4�����ʵ���Ϊ mol��
�۴�����������ȫ���ͷź�����Һ�еμ�VmL a mol��L-1NaOH��Һ��ǡ��ʹ��Һ��
��Cu2+ȫ��ת��Ϊ��������ԭŨ������H2SO4�����ʵ���Ũ��= mol��L-1��
��1��
 ��2��
��2��
��3����ɫʯ����Һ��� ��4��bd ��5���� 5.6���� 0.25���ۣ�aV��10-2+5��
�����������1����ͼʱBװ���е����ܳ����̳�����2��ͭ��Ũ���ᷴӦ���ɶ������������ͭ��ע����ƽ����3��������������ˮ���õ��ĵ���Һ������ ����ɫʯ����Һ��죻��4���ܹ�������֤��Ӧֹͣ�����ƿ��ȷ��ʣ���H2SO4����b��d��a.��Ӧ�����ɵ�����ͭ��Ҳ������������ӣ�����BaCl2��Һ���ɰ�ɫ���������ܼ��������Ĵ��ڣ�b�������Һ��������ʣ�࣬����Ba(NO3)2��Һ���������ɰ�ɫ���ᱵ��������������ƿ�ڴ��۲쵽����ɫ��NO2���壬���ܼ��������Ĵ��ڣ�c����������û�����Ա仯�����ܼ��������Ĵ��ڣ�d�������Һ��������ʣ�࣬����Na2CO3��ĩ��������ɫ��CO2���壬���ܼ��������Ĵ��ڣ���5���ٸ���ͭ������������ͭ�����ᷴӦ�Ļ�ѧ����ʽ���㣻�ڸ���Ԫ���غ㣬����ԭ��H2SO4�����ʵ�����������SO2�����ʵ������ۡ�ǡ��ʹ��Һ�е�Cu2+ȫ��ת��Ϊ�����������ʱ����ҺΪNa2SO4��Һ��������Ԫ���غ㣬����������ʵ�����n��H2SO4��=n��Na2SO4��+n��SO2��=n��NaOH����1/2+n��SO2����Ȼ�����c=n/V(aq)���㡣

��ϰ��ϵ�д�
�����Ŀ
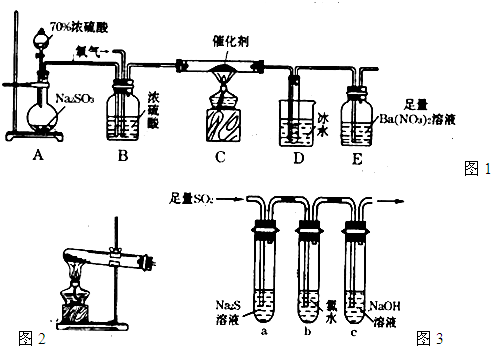
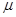 L/L����Ҫ���Դ���������ų�����������֮һ���ð�ˮϴ�����������÷���ʽ��ʾ��ˮϴ���������ų���β���Ļ�ѧ����ʽ
L/L����Ҫ���Դ���������ų�����������֮һ���ð�ˮϴ�����������÷���ʽ��ʾ��ˮϴ���������ų���β���Ļ�ѧ����ʽ 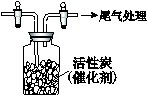
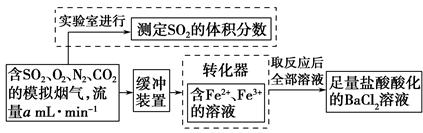
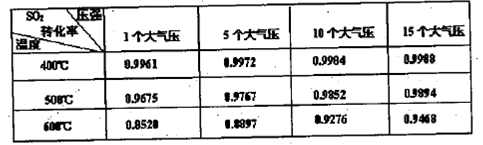
 ��ת���ʣ���֪�������٣�����ⶨ�������� �� ��
��ת���ʣ���֪�������٣�����ⶨ�������� �� ��