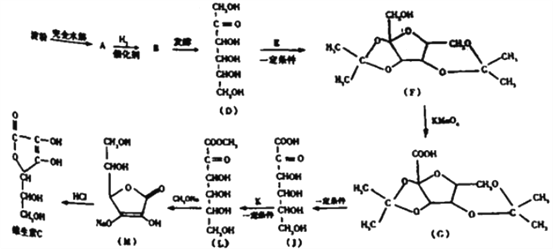
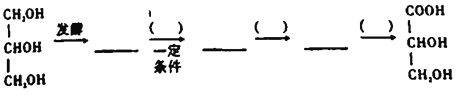��Ŀ����
����Ŀ��Ϊ���о���ѧ��ӦA+B=C+D�������仯�����ijͬѧ�������ͼ��ʾװ�á�����ʢ��A���Թ��еμ��Լ�Bʱ������U�ι��м״�Һ���½��Ҵ�Һ��������

�Իش��������⣺
��1���÷�ӦΪ_______��Ӧ(����ȡ������ȡ�)��
��2��A��B����������C��D��������_______(��ߡ��͡�)��
��3����Ӧ�ﻯѧ���������յ�����_______(��ߡ��͡�)�������ﻯѧ���γɷų���������
���𰸡����� �� ��
��������
����ʢ��A���Թ��еμ��Լ�Bʱ������U���м״�Һ���½��Ҵ�Һ����������������������������ʣ���֪�÷�ӦΪ���ȷ�Ӧ��
(1)���ڷ�����ӦA+B=C+D��U���м״�Һ���½��Ҵ�Һ��������������������������������ʿ����жϸ÷�ӦΪ���ȷ�Ӧ��
(2)����A+B=C+D�ķ�ӦΪ���ȷ�Ӧ������A��B����������C��D���������ߣ�
(3)��ѧ��Ӧ�оɼ����������������¼����ɷų��������÷�ӦΪ���ȷ�Ӧ����Ӧ�ﻯѧ���������յ��������������ﻯѧ���γɷų���������

����Ŀ���о�![]() ��
��![]() ��
��![]() �ȵĴ��������Ի�����������Ҫ���塣
�ȵĴ��������Ի�����������Ҫ���塣
��1����ѧ�������о����ô�������β���е�NO��COת���![]() ��
��![]() ���䷴ӦΪ��
���䷴ӦΪ��![]()
![]()
��Ϊ���о���������Ը÷�Ӧ��Ӱ�죬�����±�����ʵ�飬��ò�ͬʱ��NO��Ũ�ȣ�c����ʱ��仯��������ͼ1��ʾ��1��2��3������ʵ����������________������֪��ʹ�õ���������ʱ����������ȱ���������ѧ��Ӧ���ʡ���
ʵ���¶�NO��ʼŨ��O��ʼŨ�ȴ����ȱ��������������ţ��棩
ʵ�� ��� | �¶� ���棩 | NO��ʼŨ��
| CO��ʼŨ��
| �����ȱ����
| �������� ��g�� |
�� | 280 |
|
| 82 | 50 |
�� | 280 |
|
| 124 | 50 |
�� | 350 |
|
| 124 | 50 |
��ͼ2��ʾNO��ƽ��ת���ʣ�a�����¶ȡ�ѹǿ�仯��ʾ��ͼ��X��ʾ����________��������________��Y��ʾ����________����Y1________Y2������>������<������
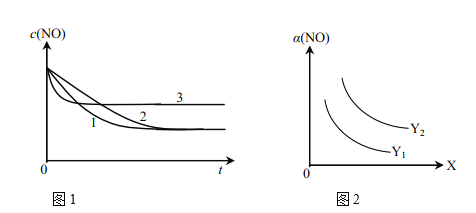
��2��һ���¶��£���![]() ��
��![]() �������1��2�����ܱ������з�����Ӧ
�������1��2�����ܱ������з�����Ӧ![]() ���ﵽƽ��ʱ
���ﵽƽ��ʱ![]() ���������Ϊ25�����÷�Ӧ��ƽ�ⳣ��
���������Ϊ25�����÷�Ӧ��ƽ�ⳣ��![]() ________��
________��
��3������ԭ��ط�Ӧ��ʵ��![]() ���������ܷ�ӦΪ
���������ܷ�ӦΪ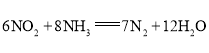 ���������ҺΪ���ԡ�����һ��ʱ��õ�ظ�����������ҺpH________�����������������С���������������������缫��ӦʽΪ________��
���������ҺΪ���ԡ�����һ��ʱ��õ�ظ�����������ҺpH________�����������������С���������������������缫��ӦʽΪ________��
����Ŀ���ش��������⣺
��1����25�桢101kPaʱ��1.00g C6H6(l)ȼ������CO2��H2O(l)ʱ���ų�41.8kJ����������ʾC6H6(l)ȼ���ȵ��Ȼ�ѧ����ʽΪ___��
��2��1.00L 1.00mol/LH2SO4��Һ��2.00L 1.00mol/L NaOH��Һ��ȫ��Ӧ���ų�114.6kJ����������ʾ���к��ȵ��Ȼ�ѧ����ʽΪ___��
��3����֪��ӦCH3-CH3��CH2=CH2(g)+H2(g)���йػ�ѧ���ļ������£�
��ѧ�� | C-H | C=C | C-C | H-H |
����E/kJ/mol | 414.4 | 615.3 | 347.4 | 435.3 |
ͨ������ó��÷�Ӧ�ķ�Ӧ��Ϊ_______________��
��4����֪2.0gȼ����(N2H4)������ȫȼ������N2��H2O(g)ʱ���ų�33.4kJ����������÷�Ӧ���Ȼ�ѧ����ʽΪ________��