��Ŀ����
�������ѣ�TiO2���㷺Ӧ���ڸ���ṹ����Ϳ�ϡ�ֽ��Ϳ��ȣ��������ѻ�����Ϊ�Ʊ��ѵ��ʵ�ԭ�ϡ�
�������ѿ����������ַ����Ʊ���
����1��TiCl4ˮ������TiO2��xH2O�����ˡ�ˮϴ��ȥ���е�Cl�����ٺ�ɡ����ճ�ȥˮ�ֵõ�����TiO2���˷����Ʊ��õ��������������ѡ�
��1���� TiCl4ˮ������TiO2��x H2O�Ļ�ѧ����ʽΪ_______________________________ ��
�ڼ���TiO2��x H2O��Cl���Ƿ����ķ�����______________________________
����2�����ú���Fe2O3����������Ҫ�ɷ�ΪFeTiO3������TiԪ�ػ��ϼ�Ϊ+4�ۣ���ȡ������Ҫ�������£�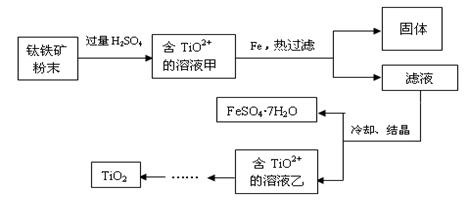
��2���������ĩ�м����ᷴӦ��TiO2+�����ӷ���ʽΪ
��3����Һ���м���Fe��������
��4����Ҫ����FeSO4��7H2O�������ˮ�������������ƾ��ơ��������⣬��Ҫ�õ������ֹ�������������
��������ѿ�������ȡ�ѵ��ʣ��漰���IJ�������ͼ��
��Ӧ�ڵķ���ʽ�� ���÷�Ӧ��Ҫ��Ar�����н��У������ԭ�� _
��1���� TiCl4+ (x+2)H2O =TiO2��x H2O+ 4HCl
��ȡ���һ�ε�ϴ����Һ1��2 mL���Թ��У������еμ������ữ��AgNO3��Һ�����ް�ɫ���������������������ϴ�Ӹɾ���
��2��FeTiO3+ 4H+ = TiO2 + +Fe2+ + 2H2O
��3������Һ�е�Fe3+��ԭΪFe2+
��4������ ������
��TiCl4��2Mg��2MgCl2��Ti ��ֹ������Mg��Ti��������е���������CO2��N2�����ã�
�����������������1������TiCl4��ϵ��Ϊ1������Ԫ���غ㣬TiO2?xH2O��ϵ��Ϊ1��HCl��ϵ��Ϊ4���ٸ���OԪ���غ㣬��֪H2O��ϵ��Ϊ��2+x��������ʽΪTiCl4+��x+2��H2O?TiO2?xH2O��+4HCl���ʴ�Ϊ��TiCl4+��x+2��H2O?TiO2?xH2O��+4HCl���ڳ���������Һ�е�Cl-�����ݷ�����Ӧ��Cl-+Ag+�TAgCl����ȡ����ϴ��Һ��������Һ���ܽ�������Ƿ��ڣ��ʴ�Ϊ��ȡ����ˮϴҺ���μ������ữ��AgNO3��Һ����������ɫ������˵��Cl-�ѳ�������2������������ԭԭ�����������ĩ�м����ᷴӦ��TiO2+�����ӷ���ʽΪFe2O3+6H+=2Fe3++3H2O��FeTiO3+4H+=Fe2++TiO2++2H2O����3������Һ�к��������ӣ������л�ԭ�ԣ��ܽ������������������������Ҳ������µ����ʣ��������������ǣ���Fe3+ת��ΪFe2+���ʴ�Ϊ����Fe3+ת��ΪFe2+����4����Ҫ����FeSO4��7H2O�������ˮ�������������ƾ��ơ��������⣬��Ҫ�õ������ֹ������������������������ǡ���800�������£����Ȼ��Ѻ�þ��Ӧ�����Ȼ�þ���ѣ���Ӧ����ʽΪ��TiCl4+2Mg=2MgCl2+Ti��Mg�ǻ��ý�������������ж������ʷ�Ӧ����˿ɵó�Ar������Ϊ����������ֹMg�Ϳ��������ʷ�Ӧ���ʴ�ΪTiCl4+2Mg=2MgCl2+Ti�� ��ֹ������Mg��Ti��������е�O2����CO2��N2�����á�
���㣺����ѧ���Թ����������⡢�Ķ���Ŀ��ȡ��Ϣ������������ԭ��Ӧ������ʽ����д�ȣ��Ѷ��еȣ�����������ǹؼ�����Ҫѧ�������Ķ���Ŀ��ȡ��Ϣ������������û���֪ʶ�������⡢�������������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�



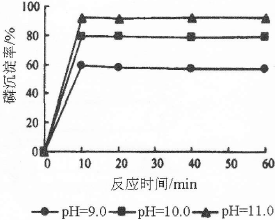

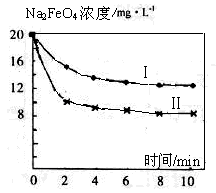

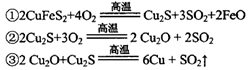
 )���Ʊ�Fe2O3���������̻ش��������⣺
)���Ʊ�Fe2O3���������̻ش��������⣺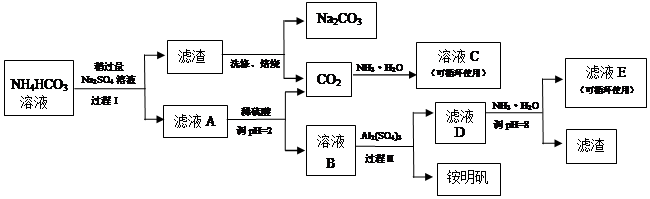

 LiF�����÷�Ӧ��ƽ�ⳣ������ʽΪK= ��
LiF�����÷�Ӧ��ƽ�ⳣ������ʽΪK= �� CO2(g)��H2(g)����H<0�������COת���ʿɲ��õķ��������У��ٽ����¶ȣ�������ѹǿ����ʹ�ô�����������CO��Ũ�ȣ�������ˮ������Ũ�ȣ�������ȷ�������(����)��
CO2(g)��H2(g)����H<0�������COת���ʿɲ��õķ��������У��ٽ����¶ȣ�������ѹǿ����ʹ�ô�����������CO��Ũ�ȣ�������ˮ������Ũ�ȣ�������ȷ�������(����)��