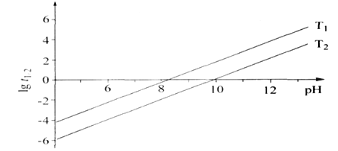
��Ŀ����
����Ŀ��������Ȼ����������̬�Ͷ��ֻ���̬��ʽ���֣���Ļ���������������Ի�ԭ�ԣ������������������ˮ�����������գ�
(1)SO2��ʹKMnO4��Һ��ɫ����ԭ������MnSO4��д���÷�Ӧ�Ļ�ѧ����ʽ__________��
(2)��SO2ͨ��BaCl2��Һ�����ͣ�δ���������ɣ��μ���ˮ���ְ�ɫ�������ð�ɫ������ѧʽΪ________��
(3)��Na2S�����зֱ����Ũ�����ϡ���ᣬ���ò���__________(������ͬ��������ͬ��)���ɴ˿�֪���������Ե�ǿ����__________�йء�
(4)��֪NaHSO3��Һ�����ԣ���Һ������Ũ���ɴ�С��˳����_____________��
(5)Ϊ��ʹNa2S��Һ��c(Na+)/c(S2)�ı�ֵ��С������������(��ͨ��)��������___________
a.H2S���� b.NaOH���� c.HCl���� d.KOH����
(6)��ͼ�У���A��ͨ��һ����SO2����B�������µ��������ɡ�B���ų���������(������������������)___________H2S��(��֪������H2SO4��H2CO3��H2S)

���𰸡�5SO2+2KMnO4+2H2O=2MnSO4+K2SO4+2H2SO4 BaSO4 ��ͬ Ũ�� [Na+]>[HSO3-]>[H+]>[SO32]>[OH] d ����
��������
��1��SO2��ʹKMnO4��Һ��ɫ����ԭ������MnSO4��+4�۵�������Ϊ+6�ۣ���Ӧ�ķ���ʽΪ��5SO2+2KMnO4+2H2O�T2MnSO4+K2SO4+2H2SO4��
��2��������������ˮ��Ӧ��������������ᣬ�������Ȼ�����Ӧ�������ᱵ��
��3����Na2S��Ũ���ᷢ��������ԭ��Ӧ���ɵ������Na2S��ϡ���ᷢ�����ֽ��������⣬���ò��ﲻͬ���ɴ˿�֪���������Ե�ǿ����Ũ���йأ�
��4����֪NaHSO3��Һ�����ԣ�˵���������ˮ�⣻
��5����������Һ�е���������Ӻ������ӣ�������ˮ�⣬����������Ũ�Ȼ��С��Ҫʹ![]() ��С�������������ˮ��ƽ���ƶ���������
��С�������������ˮ��ƽ���ƶ���������
��6��������������������з�Ӧ���ɵ��������Բ����ܺ����⡣
��1��SO2��ʹKMnO4��Һ��ɫ����ԭ������MnSO4��+4�۵�������Ϊ+6�ۣ���Ӧ�ķ���ʽΪ��5SO2+2KMnO4+2H2O�T2MnSO4+K2SO4+2H2SO4���ʴ�Ϊ��5SO2+2KMnO4+2H2O�T2MnSO4+K2SO4+2H2SO4��
��2��������������ˮ��Ӧ��������������ᣬ�������Ȼ�����Ӧ�������ᱵ���ʴ�Ϊ�� BaSO4��
��3����Na2S��Ũ���ᷢ��������ԭ��Ӧ���ɵ������Na2S��ϡ���ᷢ�����ֽ��������⣬���ò��ﲻͬ���ɴ˿�֪���������Ե�ǿ����Ũ���йأ��ʴ�Ϊ����ͬ��Ũ�ȣ�
��4����֪NaHSO3��Һ�����ԣ�˵���������ˮ�⣬��������Ũ�ȵĴ�СΪ��[Na+]��[HSO3-]��[H+]��[SO32-]��[OH-]���ʴ�Ϊ��[Na+]��[HSO3-]��[H+]��[SO32-]��[OH-]��
��5����������Һ�е���������Ӻ������ӣ�������ˮ�ⷽ��ʽΪ��S2-+H2OHS-+OH-��Ҫʹ![]() ��С����ʹ������ˮ��ƽ���淽���ƶ����ɣ����Լ�����KOH��Һ��c(OH-)����������ˮ��ƽ���淽���ƶ���c(S2-)����
��С����ʹ������ˮ��ƽ���淽���ƶ����ɣ����Լ�����KOH��Һ��c(OH-)����������ˮ��ƽ���淽���ƶ���c(S2-)����![]() ��С����d��ȷ����ѡd��
��С����d��ȷ����ѡd��
��6��������������������з�Ӧ���ɵ��������Բ����ܺ����⣬�ʴ�Ϊ��������