��Ŀ����
4�� ��ϸͭ����ҪӦ���ڵ�����ϡ������������У���ϸͭ�۵�ij
��ϸͭ����ҪӦ���ڵ�����ϡ������������У���ϸͭ�۵�ij�Ʊ��������£�Cu��NH3��4SO4��ˮ��Һ��$��_{����}^{ͨ��SO_{2}}$NH4CuSO3����ɫ������$��_{�ȡ�����}^{����10mol/L����}$��ϸͭ��
�Իش��������⣺
��1�����й���[Cu��NH3��4]SO4��˵���У���ȷ����AD��
A��[Cu��NH3��4]SO4�������Ļ�ѧ�������Ӽ������Լ�����λ��
B��[Cu��NH3��4]SO4����NH3���ӣ���ˮ��Һ��Ҳ����NH3����
C��[Cu��NH3��4]SO4�����Ԫ���е�һ��������������Ԫ��
D��[Cu��NH3��4]SO4��������ӵĿռ乹��Ϊ��������
��2��NH4CuSO3�еĽ��������ӵĺ�������Ų�ʽΪ[Ar]3d10��
��3��SO${\;}_{3}^{2-}$������S��ԭ�ӵ��ӻ���ʽΪsp3�����以Ϊ�ȵ������һ�ַ��ӵķ���ʽ��NF3����PF3��NCl3��PCl3�ȣ���
��4��NH3��Һ����ԭ����NH3���Ӽ���γ������
��5����ͼ��ͭ��ij��������ľ����ṹʾ��ͼ���ɴ˿�ȷ����������Ļ�ѧʽΪCuO��
��6��NH4CuSO3�������ȷ�Ӧ�����ӷ���ʽΪ2NH4CuSO3+4H+$\frac{\underline{\;��\;}}{\;}$2NH4++Cu2++Cu+2SO2��+2H2O��
���� ��1��A���������Ӵ������Ӽ����ǽ���Ԫ�ؼ����γɹ��ۼ���������д�����λ����
B��NH3Ϊ������ӣ���Һ�в�����NH3��
C��������������ɼ���������жϣ�
D�����ݼ۲���ӶԻ��������ж����ӹ��ͣ�
��2��NH4CuSO3��Cu�Ļ��ϼ�Ϊ+1�ۣ���̬Cu+������28�����ӣ����ݹ���ԭ����д���������Ų�ʽ��
��3��SO32-�к���3���ļ���1���µ��Ӷԣ���ϵȵ�����Ķ����жϣ�
��4�����ڰ�����֮����������ڣ��������ӵķе�ߣ���Һ����
��5�����þ�̯�����㣻
��6�����õ���ϸͭ���⣬���ɿ�����Һ������ͬʱ��ʹƷ����ɫ�����壬˵������Cu��Cu2+��SO2��
��� �⣺��1��A��[Cu��NH3��4]SO4����������Ӻ�[Cu��NH3��4]2+�������Ӽ���Nԭ�Ӻ�ͭԭ��֮�������λ����NH3��H��N֮����ڹ��ۼ�������[Cu��NH3��4]SO4�������Ļ�ѧ���й��ۼ������Ӽ�����λ������A��ȷ��
B��NH3Ϊ������ӣ���Һ�в�����NH3����B����
C��������������ɿ����ж�������NH3����Nԭ������߹¶Ե��ӣ�����ԭ�����ṩ�չ������C����
D��SO42-�����м۲���Ӷ���Ϊ4+$\frac{1}{2}$����6+2-4��2��=4���µ��Ӷ���Ϊ0�������������������ͣ���D��ȷ��
�ʴ�Ϊ��AD��
��2��Cuԭ�ӵĺ�������Ų�ʽΪ[Ar]3d104s1��NH4CuSO3��Cu�Ļ��ϼ�Ϊ+1�ۣ���̬Cu+������28�����ӣ����ݹ���ԭ�����������ӵĺ�������Ų�ʽΪ[Ar]3d10��
�ʴ�Ϊ��[Ar]3d10��
��3��SO32-�к���3���ļ���1���µ��Ӷԣ���Ϊsp3�ӻ������Ӻ���4��ԭ�ӣ��۵�������Ϊ26����Ӧ�ĵȵ�������NF3����PF3��NCl3��PCl3�ȣ���
�ʴ�Ϊ��sp3��NF3����PF3��NCl3��PCl3�ȣ���
��4��������֮�������γ������ʹ��е����߶�����Һ����
�ʴ�Ϊ��������֮�������γ������ʹ��е����߶�����Һ����
��5��������4��Cuλ�ھ����ڲ���Oλ�ھ����Ķ�������ģ�����8��$\frac{1}{8}$+6$\frac{1}{2}$=4�����������Ļ�ѧʽΪCuO��
�ʴ�Ϊ��CuO��
��6�����õ���ϸͭ���⣬���ɿ�����Һ������ͬʱ��ʹƷ����ɫ�����壬˵������Cu��Cu2+��SO2����Ӧ�����ӷ���ʽΪ2NH4CuSO3 +4H+$\frac{\underline{\;��\;}}{\;}$2NH4++Cu2++Cu+2SO2��+2H2O��
�ʴ�Ϊ��2NH4CuSO3 +4H+$\frac{\underline{\;��\;}}{\;}$2NH4++Cu2++Cu+2SO2��+2H2O��
���� �����Ϊ�ۺϣ�ͻ�����������֪ʶ����������Ų����ӻ����͡���ѧ�����������ʡ������ɼ�״�����˽⣬�ۺ��Խ�ǿ��ȫ�濼�����ʵĽṹ�����ʣ���Ŀ�ѶȽϴ�

 ��ѧ��������������Ͼ���ѧ������ϵ�д�
��ѧ��������������Ͼ���ѧ������ϵ�д� �ϴ�̸�������������νӽ̳��Ͼ���ѧ������ϵ�д�
�ϴ�̸�������������νӽ̳��Ͼ���ѧ������ϵ�д�| A�� | �����������ᷴӦ | B�� | ľ̿��Ũ���ᷴӦ | ||
| C�� | ͭ�����ᷴӦ | D�� | �������������ᷴӦ |
| A�� | 3.01��1023 | B�� | 6.02��1023 | C�� | 0.5 | D�� | 1 |
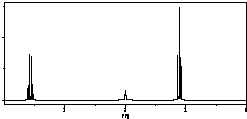
| A�� | CH3CH2OH | B�� |  | C�� | 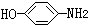 | D�� | 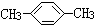 |
| A�� | ��Һ�е������ӵ���Ŀ���� | |
| B�� | ��Һ��$\frac{c��C{H}_{3}CO{O}^{-}��•c��{H}^{+}��}{c��C{H}_{3}COOH��}$���� | |
| C�� | ����ĵ���̶�����c��H+�������� | |
| D�� | �ټ���10mL pH=11��NaOH��Һ�����ҺpH=7 |
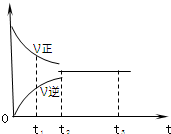 ��ͼ�ǿ��淴ӦX2+3Y2?2Z2 �ڷ�Ӧ�����еķ�Ӧ���ʣ�V����ʱ�䣨t���Ĺ�ϵ���ߣ�����������ȷ���ǣ�������
��ͼ�ǿ��淴ӦX2+3Y2?2Z2 �ڷ�Ӧ�����еķ�Ӧ���ʣ�V����ʱ�䣨t���Ĺ�ϵ���ߣ�����������ȷ���ǣ�������| A�� | t1ʱ������Ӧ����С���淴Ӧ���� | B�� | t2ʱ������Ӧ���ʵ����淴Ӧ���� | ||
| C�� | t2-t3���ÿ��淴Ӧ���ٷ��� | D�� | t2-t3�������ʵ�Ũ�Ȳ��ٷ����仯 |
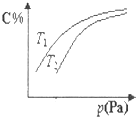 ���ܱ������У����ڿ��淴Ӧ2A+3B?2C��g�� ƽ��ʱC������������¶Ⱥ�ѹǿ�Ĺ�ϵ��ͼ��ʾ������˵������ȷ���ǣ�������
���ܱ������У����ڿ��淴Ӧ2A+3B?2C��g�� ƽ��ʱC������������¶Ⱥ�ѹǿ�Ĺ�ϵ��ͼ��ʾ������˵������ȷ���ǣ�������| A�� | ������Ӧ�����H��0����T2��T1 | |
| B�� | ѹǿ����ʱ����������ƽ����Է����������� | |
| C�� | A��һ��Ϊ���� | |
| D�� | Bһ��Ϊ���� |
| ��ѧ�� | H-H | O=O |
| ����/KJ?mol-1 | 436 | 498 |
| A�� | 463.4KJ/mol | B�� | 926.8KJ/mol | C�� | 221.6KJ/mol | D�� | 413KJ/mol |
 ���ڱ�ǰ�����ڵ�Ԫ��a��b��c��d��e��ԭ��������������A�ĺ����������������Ӳ�����ͬ��b�ļ۵��Ӳ��е�δ�ɶԵ�����3����c������������Ϊ���ڲ��������3����d��cͬ���壬e�������ֻ��1�����ӣ����������18�����ӣ��ش��������⣺
���ڱ�ǰ�����ڵ�Ԫ��a��b��c��d��e��ԭ��������������A�ĺ����������������Ӳ�����ͬ��b�ļ۵��Ӳ��е�δ�ɶԵ�����3����c������������Ϊ���ڲ��������3����d��cͬ���壬e�������ֻ��1�����ӣ����������18�����ӣ��ش��������⣺ ��
��