��Ŀ����
4���ݶ�����������SK-��ٻ�̡���ޢ���ϰ¡���ʫ���컯ױƷ�еķ۱���������и���Υ�����ʣ���λ�����ڱ��е���ͬ�ڢ�B�壬����Ҫ���ϼ�Ϊ+6��+3��+2������+3�۽�Ϊ�ȶ���ijУ��ѧ��ȤС��Ը��Ļ��������̽�����о�����1��Cr2O3����ˮ����Al2O3���ƣ������������Cr��OH��3��Al��OH��3���ƣ�Ҳ���������������ˮ�д�����ʽ�ͼ�ʽ���룬����뷽��ʽΪCrO2-+H++H2O?Cr��OH��3?Cr3++3OH-��
��2����ҵ�Ϻ�ʵ�����г����ĸ���+6�ۣ����������京�����Σ�������κ��ظ����Σ���K2Cr2O7��Na2Cr2O7��Ϊ��Ҫ����K2Cr2O7��Һ�д�����Cr2O72-��CrO42-�����ﵽ����ƽ�⣺Cr2O72-+H2O?2CrO42-+2H+����H+Ũ������ʱ��ƽ��������Cr2O72-�ķ����ƶ�����Һ�ʳȺ�ɫ����OH-Ũ������ʱ����������H+����ʹƽ��������CrO42-�ķ����ƶ�����Һ�ʻ�ɫ��
���� ��1���������������ĵ��뷽��ʽ��дCr��OH��3�ĵ��뷽��ʽ��
��2��Cr2O72-+H2O?2CrO42-+2H+����H+Ũ������ʱ��ƽ�����淴Ӧ�ķ����ƶ�����OH-Ũ������ʱ����������H+����ʹƽ��������Ӧ�ķ����ƶ����ݴ˷�����
��� �⣺��1���������������ĵ��뷽��ʽ��дCr��OH��3�ĵ��뷽��ʽΪCrO2-+H++H2O?Cr��OH��3?Cr3++3OH-���ʴ�Ϊ��CrO2-+H++H2O?Cr��OH��3?Cr3++3OH-��
��2��Cr2O72-+H2O?2CrO42-+2H+����H+Ũ������ʱ��ƽ�����淴Ӧ�ķ����ƶ���������Cr2O72-�ķ����ƶ���������Һ�ʳȺ�ɫ����OH-Ũ������ʱ����������H+����ʹƽ��������Ӧ�ķ����ƶ��������� CrO42-�ķ����ƶ���������Һ�ʻ�ɫ���ʴ�Ϊ��Cr2O72-���Ⱥ죻CrO42-���ƣ�
���� ���⿼�黯ѧƽ���ƶ���Ӱ�죬ע��Cr2O72-+H2O?2CrO42-+2H+��ƽ���ƶ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
15�����з�Ӧ������CO2���������ǣ�������
| A�� | O2+C$\frac{\underline{\;����\;}}{\;}$CO2 | B�� | 2NaOH+CO2�TNa2CO3+H2O | ||
| C�� | 2Na2O2+CO2�T2Na2CO3+O2 | D�� | 2Mg+CO2$\frac{\underline{\;��ȼ\;}}{\;}$2MgO+C |
19��ԭ����������11-35֮�����������Ԫ��A��B�����ӷֱ�ΪAm+��Bn-����д�±�
| λ�� | A��B��������� | ���Ӱ뾶�Ƚ� | ||||
| ���� | ���� | ���� | ||||
| Am+��Bn-����������Ӳ� | A | |||||
| B | ||||||
| Am+��Bn-���һ�����Ӳ� | A | |||||
| B | ||||||
| Am+��Bn-���Ӳ�ṹ��ͬ | A | |||||
| B | ||||||
16����9.0gͭ�����Ļ����Ͷ��100mlϡ�����г�ַ�Ӧ�õ������1.12LNO��ʣ��4.8g��������������100ml��Ũ�ȵ�ϡ���������ȫ�ܽ⣬�ֵõ������1.12LNO������Ӧ�����Һ�е���KSCN��Һ����죬������˵����ȷ���ǣ�������
| A�� | ��һ����100mlϡ���ᷴӦʣ4.8g����ΪCu��Fe | |
| B�� | ��Ӧǰϡ��������ʵ�����Ũ��Ϊ1.5 mol•L-1 | |
| C�� | �����������������Һ���ټ���������ϡ���ỹ�ɵõ������0.56LNO | |
| D�� | ԭ�������Cu��Fe�����ʵ����ֱ�Ϊ0.06mol��0.09mol |
6����֪������Cu��OH��2���ܶȻ�Ϊ2.2��10-22�������£���0.20mol/LCuSO4��Һ�м���NaOH��Һ����ֽ��裬��dz��ɫ����Cu��OH��2���ɣ�����Һ��pH=10ʱ����Һ�е�Cu2+�����ʵ���Ũ��Ϊ��������
| A�� | 2.2��10-10 mol/L | B�� | 2.2��10-12 mol/L | C�� | 2.2��10-14 mol/L | D�� | 1.1��10-21 mol/L |
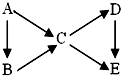 AΪ���ʣ�B��C��D��EΪ��A������ͬԪ�صĻ��������֮��������ת����ϵ��
AΪ���ʣ�B��C��D��EΪ��A������ͬԪ�صĻ��������֮��������ת����ϵ��
 ����ˮ��Һ�����ڿ����з������ױ���ǣ��ñ仯���ֳ���S�ǽ����Ա�O��O���ǿ��������������ԭ�ӽṹ����ԭ��ͬ����Ԫ��������������ͬ�����ϵ��£����Ӳ������࣬ԭ�Ӱ뾶���õ�������������
����ˮ��Һ�����ڿ����з������ױ���ǣ��ñ仯���ֳ���S�ǽ����Ա�O��O���ǿ��������������ԭ�ӽṹ����ԭ��ͬ����Ԫ��������������ͬ�����ϵ��£����Ӳ������࣬ԭ�Ӱ뾶���õ�������������