��Ŀ����
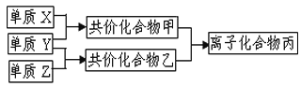
����Ŀ��������Ԫ�صĵ���X��Y��Z��ͨ��״���¾�Ϊ��̬��������ͼת����ϵ����Ӧ������ȥ������֪X��Y��Z��Ϊ˫ԭ�ӵ��ʣ�X�ǿ����к����������壬��ʹʪ��ķ�̪��ֽ��죬������ˮ�������ᡣ������������⣺
��1��X�ĵ���ʽ��_________________��
��2��д�����ҷ�Ӧ�Ļ�ѧ����ʽ__________________��
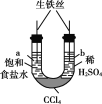
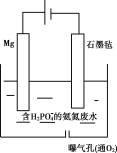
��3��ʵ���ҿ�����ͼ��ʾ��װ�ã�ȱ���ռ�װ�ã��г̶ֹ�װ����ȥ���Ʊ����ռ��ס�
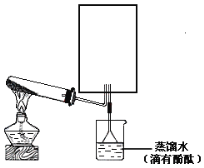
����ͼ�з����ڻ���ռ�������װ�ü�ͼ��_______________
���Թ��е��Լ���________________����д��ѧʽ����
���ձ�����Һ����ɫ��Ϊ��ɫ����ԭ���ǣ��õ��뷽��ʽ��ʾ��_________________________________��
���𰸡�![]() NH3+HCl=NH4Cl
NH3+HCl=NH4Cl  ���������֣� Ca(OH)2��NH4Cl NH3H2O NH4++OH�D
���������֣� Ca(OH)2��NH4Cl NH3H2O NH4++OH�D
��������
��֪X��Y��Z��Ϊ˫ԭ�ӵ��ʣ�X�ǿ����к����������壬��XΪ��������ʹʪ��ķ�̪��ֽ��죬���Ϊ������������ˮ�������ᣬ����ΪHCl������YΪ����������ZΪ������
��1��XΪ�����������ʽΪ![]() ��
��
��2�����ҷֱ�Ϊ������HCl�����ʱ��Ӧ�����Ȼ�泥���ѧ����ʽΪNH3+HCl=NH4Cl��
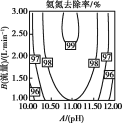
��3���ټ�Ϊ�������ܶȱȿ���С����������ˮ����ֻ���������ſ������ռ�����װ��Ϊ ��
��
��ʵ����ͨ�����Ȼ������ʯ�һ�ϼ�����ȡ�������Լ�ΪCa(OH)2��NH4Cl��
�۰�����ˮ������һˮ�ϰ�����Ϊ���ʹ��̪��Һ��죬�������笠����Ӻ����������ӣ����뷽��ʽΪNH3H2O NH4++OH�D��

 Сѧ��ʱ��ѵϵ�д�
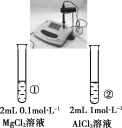
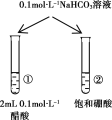
Сѧ��ʱ��ѵϵ�д�����Ŀ������ʵ���������������Ӧ���ǣ� ��
A | B | C | D | |
ʵ�� |
|
|
|
|
���� | һ��ʱ���a��Һ�����b��Һ�� | ����KMnO4��Һ��ɫ | pH�Ʋ�â���pH���ڢ���pH | �Թܢ����д������ݣ��Թܢ��������� |
���� | a�ܷ���������ʴ��b�ܷ������ⸯʴ | ����ϩ���ɿ���ʹ����KMnO4��Һ��ɫ | �����ԣ�Mg��Al | ���ԣ����̼����� |
A.AB.BC.CD.D