��Ŀ����
3��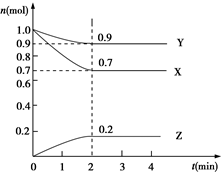 ij�¶�ʱ����2L���ܱ������У�X��Y��Z�������ʣ�����XΪ���壬Y��ZΪ���壩�����ʵ�����ʱ��ı仯������ͼ��ʾ��
ij�¶�ʱ����2L���ܱ������У�X��Y��Z�������ʣ�����XΪ���壬Y��ZΪ���壩�����ʵ�����ʱ��ı仯������ͼ��ʾ����1����ͼ���������ݽ��з������÷�Ӧ�Ļ�ѧ����ʽΪX+3Y?2Z��
��2����Ӧ�ӿ�ʼ��2����ĩ����Z��Ũ�ȱ仯��ʾ��ƽ����Ӧ����Ϊv��Z��=0.05mol•��L•min��-1��
��3���������������䣬ֻ�ı�һ����Ӧ����������Ӧ���ʵı仯������пո��
������ĸ��A������ B����С C�����䣩
| �ı����� | �����¶� | ����X���� | ʹ�ô��� | ����������� |
| ���ʱ仯 |
���� ��1���������ʵ����ı仯�жϷ�Ӧ���������������ʵ����ı仯֮�ȵ��ڻ�ѧ������֮����д����ʽ��
��2������v=$\frac{��c}{��t}$���㷴Ӧ���ʼ��ɣ�
��3������Ӱ�컯ѧ��Ӧ���ʵ����أ��¶ȡ�Ũ�ȡ�ѹǿ������֪ʶ���ش�
��� �⣺��1����ͼ����Կ�������Ӧ��X��Y�����ʵ�����С��Z�����ʵ������࣬��X��YΪ��Ӧ�ZΪ�����
�ҡ�n��X������n��Y������n��Z��=0.1mol��0.3mol��0.2mol=1��3��2����Ӧ�Ļ�ѧ����ʽΪ��X+3Y?2Z��
�ʴ�Ϊ��X+3Y?2Z��
��2��v��Z��=$\frac{\frac{0.2mol}{2L}}{2min}$=0.05mol•��L•min��-1���ʴ�Ϊ��0.05mol•��L•min��-1��
��3�����ڷ�Ӧ��X+3Y?2Z�������¶ȣ���ѧ��Ӧ���ʼ��������ӷ�Ӧ���Ũ�ȣ���Ӧ���ʼӿ죬����A�ǹ��壬�����������ʵı仯�����������Ӧ���ʼӿ죬ѹ�����������ѹǿ����Ӧ���ʼӿ죬�ʴ�Ϊ��B��C��A��A��
���� ���⿼�黯ѧƽ��ͼ�������Ŀ����Ŀ�ѶȲ���ע����������ʵ����ı仯���ߣ����ջ�ѧ����ʽ���жϷ����Լ�����Ӱ�컯ѧ��Ӧ���ʵ������ǹؼ���

��ϰ��ϵ�д�
�����Ŀ
11�����и���ʵ����ʵ���ý��ۻ������ȷ���ǣ�������
| ʵ����ʵ | ���ۻ���� | |
| A | SO2��ˮ��Һ���� | SO2�ǵ���� |
| B | AlCl3��Һ���� | AlCl3�����ӻ����� |
| C | H2SO4��Һ�����Ա�CH3COOH��Һǿ | H2SO4��ǿ����� |
| D | 0.1mol/LCH3COOH��Һ��pH��1 | CH3COOH?H++CH3COO- |
| A�� | A | B�� | B | C�� | C | D�� | D |
18������˵����ȷ���ǣ�������
| A�� | ������һ�����ڹ��ۼ� | |
| B�� | ��ԭ�ӹ��ɵľ���һ����ԭ�Ӿ��� | |
| C�� | ���������ӵĻ�����һ������������ | |
| D�� | �ǽ���Ԫ����ɵĻ�����һ���ǹ��ۻ����� |
15�����л���������Ԫ�ػ��ϼ���-1�ۻ�+1�۵��ǣ�
| A�� | KClO3 | B�� | CaCl2 | C�� | NaClO | D�� | Ca��ClO��2 |
12�����и�ʵ���У�һ��������ֳ���������ǣ�������
| A�� | ��CO2ͨ��CaCl2��Һ�� | B�� | ��H2S��ͨ�˵�Ũ������ | ||
| C�� | ��SO2ͨ��Ba��0H��2��Һ�У� | D�� | �����᳤��¶���ڿ��� |
9�����������У����ڴ�������ǣ�������
| A�� | �ྻ�Ŀ��� | B�� | ��������ˮ | C�� | ʯ�� | D�� | ��ˮ |