��Ŀ����
����Ŀ��I ��0.1 mol��þ�������������100mL 2mol��L-1��H2SO4��Һ�У�Ȼ���ٵμ�1mol��L-1��NaOH��Һ����ش𣺣�����Ҫд��������̣�
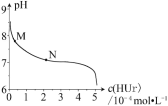
��1�����ڵμ�NaOH��Һ�Ĺ����г�������![]() �����NaOH��Һ�����V�仯��ͼ��ʾ����
�����NaOH��Һ�����V�仯��ͼ��ʾ����![]() mLʱ���������ĩ��n(Mg)=________mol��
mLʱ���������ĩ��n(Mg)=________mol��![]() _______mL��
_______mL��
��2����Ҫ����100 mL 2mol��L![]() ��H2SO4��Һ����Ҫ����������Ͳ���ձ��⣬����______________
��H2SO4��Һ����Ҫ����������Ͳ���ձ��⣬����______________
�����в�����ʹ�������ҺŨ��ƫС����___________������ţ�
A������Ͳ��ȡһ�������98%��ŨH2SO4��ϡ�ͺ�δ����ȴ��ת������ƿ��
B��ϡ���������õ�С�ձ�δϴ��
C������ʱ����Һ��
D��������ˮϴ�Ӻ������ƿδ����
E������ҡ�Ⱥ��������Լ�ƿ�д���ʱ����������Һdz��ƿ��
II��1����100 g���ʵ���Ũ��Ϊcmol/l���ܶ�Ϊpg/ mL�������м���һ������ˮϡ�ͳ�0.5cmol/L�����ᣬ�����ˮ�����________100 mL (�� ��== ����< �� �� >�� ��ͬ��
��2�����ѣ�1���е�����ijɰ�ˮ��Ӧ����ˮ�����______100 mL
��3�����ѣ�1������2�������ʵ�����Ũ�Ⱦ���Ϊ���ʵ����������������ˮ�����_____100 mL
���𰸡�0.06 440 ��������100mL����ƿ����ͷ�ι� B < > =
��������
I (1)����ͼ�����֪��,�ڵμ�NaOH��Һ�����V1=160mL�����У�û�г������ɣ�˵��������ʣ�࣬�μӵ�NaOH�����к�ʣ�����ᣬV1=160mLʱ��ʣ���H2SO4��μӵ�NaOHǡ����ȫ��Ӧ����Һ��MgSO4��Al2��SO4��3��Na2SO4�Ļ��Һ������Mgԭ�ӡ�Alԭ�ӡ�SO42-���ӡ�Na+�����غ㣬�з�������n(Mg)�����μ�NaOH��Һ�����V2ʱ��Al��OH��3��ȫ�ܽ⣬������Mg��OH��2����Һ��Na2SO4��NaAlO2���Һ������SO42-���ӡ�Na+���Ӻ�Alԭ���غ㣬���n��NaOH����������V=n/c���㣻
(2)�ٸ���������Һ��ʵ���������ѡ�������������
�ڷ������������ʵ����ʵ��������Һ�������Ӱ�죬����c=n/V�����жϣ�
II��1��ϡ��ǰ�����ʵ����ʵ������䣬��ϡ�ͺ�����Ϊԭ������Һ�����2����ϡ�ͺ�������Һ���ܶȼ�С����ϡ�ͺ���Һ������С��200g���ʼ���ˮ������С��100�ˣ��ٸ���V=m/�������ж�ˮ�������
��2����������ijɰ�ˮ��ϡ�ͺ�ˮ���ܶ�������ϡ�ͺ���Һ����������200�ˣ��ʼ���ˮ����������100�ˣ��ٸ���V=m/�������ж�ˮ�������
��3�����ѣ�1������2�������ʵ�����Ũ�Ⱦ���Ϊ���ʵ�������������ϡ�ͺ���Һ����������200�ˣ��ʼ���ˮ������100�ˣ��ٸ���V=m/�������ж�ˮ�������
(1)��V1=160mLʱ����ʱ��Һ��MgSO4��Al2��SO4��3��Na2SO4���Һ����Na+�����غ����֪��n��Na2SO4��=1/2n��Na+��=1/2n��NaOH��=1/2��0.16L��1mol/L=0.08mol����MgSO4�����ʵ���Ϊxmol��Al2��SO4��3Ϊymol����:����Mgԭ�ӡ�Alԭ���غ���:(1)x+2y=0.1������SO42-�����غ���:(2)x+3y=0.2-0.08����������(1)(2)����ó�:x=0.06��y=0.02�����Խ�����ĩ��: n(Mg)=0.06mol��n(Al)=2y=2��0.02=0.04mol���μ�NaOH��Һ�����V2ʱ����Һ��Na2SO4��NaAlO2���Һ������SO42-���ӡ�Na+���Ӻ�Alԭ���غ���:n��NaOH��=2n��Na2SO4��+n��NaAlO2��=2n��H2SO4��+n��Al��=2��0.1L��2mol/L+0.04mol=0.44mol������V2=0.44mol/1mol/L=0.44L=440mL��
����: 0.06��440��
(2)���Ʋ�����:���㡢��ȡ��ϡ�͡���Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ����ǩ�Ȳ�����һ������Ͳ��ȡ(���轺ͷ�ι�)�����ձ����ܽ⣬��ȴ��ת�Ƶ�100 mL ����ƿ�У����ò�����������ϴ�Ӳ���ϴ��Һ��������ƿ������ˮ��Һ�����̶���1-2cmʱ�����ý�ͷ�ιܵμӣ�ʹ��Һ�İ�Һ�����͵��������ƽ����ҡ�Ⱥ�,װƿ����ǩ��������Ҫ������Ϊ�����������ձ�����ͷ�ιܡ���Ͳ��100 mL����ƿ�������л���Ҫ��������:��������100 mL����ƿ����ͷ�ιܣ�
����:��������100 mL����ƿ����ͷ�ιܣ�
(2)A������Ͳ��ȡһ�������98%��Ũ���ᣬϡ�ͺ�δ����ȴ��ת������ƿ�ڣ�����Һ�����ƫ��һ����ȴ��������Һ�����ƫС��Ũ��ƫ��A����
B��ϡ���������õ�С�ձ�δϴ�ӣ����ձ��ڱ�մ�����ʣ��������ʵ�����ƫС��Ũ��ƫС����B��ȷ��
C������ʱ���ӹ۲�Һ�棬��Һ��Һ����ڿ̶��ߣ���Һ�����ƫС��Ũ��ƫ��C����
D��������ˮϴ�Ӻ������ƿδ�������Һ�������Ӱ�죬Ũ�Ȳ��䣬��D����
E������ҡ�Ⱥ��������Լ�ƿ�д���ʱ����������Һ����ƿ�⣬��Ϊ�Ǿ��ȵ���Һ������Һ��Ũ����Ӱ�죬��E����
����:B��
II��1����Ũ��Ϊcmol/l����Һϡ�ͳ�0.5cmol/L��ϡ��ǰ�����ʵ����ʵ������䣬��ϡ�ͺ���Һ�����Ϊԭ������Һ�����2����ϡ�ͺ�������Һ���ܶȼ�С����ϡ�ͺ���Һ������С��200g���ʼ���ˮ������С��100g�������ˮ�����С��100mL��
���ǣ�<��
��2����������ijɰ�ˮ��ϡ�ͺ�ˮ���ܶ�������ϡ�ͺ���Һ����������200g���ʼ���ˮ����������100g�������ˮ���������100mL��
���ǣ�>��
��3�����ѣ�1������2�������ʵ�����Ũ�Ⱦ���Ϊ���ʵ�������������ϡ�ͺ���Һ����������200g���ʼ���ˮ������100g�������ˮ���������100mL��
���ǣ�=��

 ȫ��������ϵ�д�
ȫ��������ϵ�д� һ��һ����ʱ���ϵ�д�
һ��һ����ʱ���ϵ�д�����Ŀ�����и������ʣ����ܰ� ![]() (��������ʾ��Ӧһ�����)��ϵת�������� ��
(��������ʾ��Ӧһ�����)��ϵת�������� ��
ѡ�� | a | b | c |
A | Fe | FeCl3 | FeCl2 |
B | NaOH | NaHCO3 | Na2CO3 |
C | Al | Al(OH)3 | Al2O3 |
D | MgCl2 | Mg(OH)2 | MgO |
A. A B. B C. C D. D