��Ŀ����
14����֪��A��B��F�Ǽ�ͥ�г������л��E��ʯ�ͻ�����չˮƽ�ı�־��������ͼ��ʾ��ת����ϵ�ش����⣺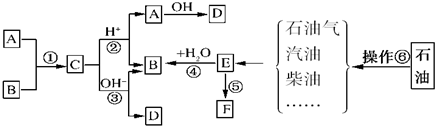
��1����������������
��2��ȡ����Ӧ�ķ���ܹ㣬�١���������ȡ����Ӧ���Ǣ٢ڢۣ�������ţ�
��3����д�����з�Ӧ�Ļ�ѧ����ʽ��
��CH2�TCH2+H2O$\stackrel{����}{��}$CH3-CH2OH��
��nCH2=CH2$\stackrel{����}{��}$
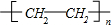 ��
����4��F��һ�ֳ����ĸ߷��Ӳ��ϣ��������Ǵ����˾�ķ��㣮Ȼ�������ֲ�����ɵĵ����ijһ���������ǰ�ɫ��Ⱦ��
���� A��B��F�Ǽ�ͥ�г������л��E��ʯ�ͻ�����չˮƽ�ı�־����EΪCH2=CH2��Eͨ���Ӿ۷�Ӧ�õ������ĸ߷��Ӳ���F����FΪ����ϩ��E��ˮ�����ӳɷ�Ӧ����BΪC2H5OH��A��B��Ӧ����C��C��������������ˮ������A��B��D����AΪCH3COOH��CΪCH3COOCH2CH3��DΪCH3COONa���ɲ�����֪��Ϊʯ�ͷ����Դ������
��� �⣺A��B��F�Ǽ�ͥ�г������л��E��ʯ�ͻ�����չˮƽ�ı�־����EΪCH2=CH2��Eͨ���Ӿ۷�Ӧ�õ������ĸ߷��Ӳ���F����FΪ����ϩ��E��ˮ�����ӳɷ�Ӧ����BΪC2H5OH��A��B��Ӧ����C��C��������������ˮ������A��B��D����AΪCH3COOH��CΪCH3COOCH2CH3��DΪCH3COONa��
��1����ʯ���и���ֵķе㲻ͬ�������÷���ķ������õ�����֣����Բ�����Ϊ����
�ʴ�Ϊ������
��2��������������֪���٢ڢ�����ȡ����Ӧ����ˮ�ӳɷ�Ӧ�������ڼӾ۷�Ӧ��
�ʴ�Ϊ���٢ڢۣ�
��3����Ӧ�ܵĻ�ѧ��Ӧ����ʽΪ��CH2�TCH2+H2O$\stackrel{����}{��}$CH3-CH2OH����Ӧ�ݵĻ�ѧ��Ӧ����ʽΪ��nCH2=CH2$\stackrel{����}{��}$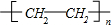 ��
��
�ʴ�Ϊ��CH2�TCH2+H2O$\stackrel{����}{��}$CH3-CH2OH��nCH2=CH2$\stackrel{����}{��}$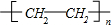 ��
��
��4��FΪ����ϩ���ϣ����Խ��⣬��ɰ�ɫ��Ⱦ��
�ʴ�Ϊ����ɫ��Ⱦ��
���� ���⿼���л�����ƶϣ�EΪ��ϩ�ǽ�����ͻ�ƿڣ��ٸ��ݸ�����֮���ת���Ƴ������ʣ���Ŀ�Ѷ��еȣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | 0.5 L1mol/L NaHS��Һ�У�N��Na+��+N��HS-��=NA | |
| B�� | 12.4g���ף�����ʽΪP4���к���P-P���ۼ�0.6NA | |
| C�� | �����£�pH=2�Ĵ�����Һ�������е�H+��Ϊ0.01NA | |
| D�� | ��92gN2O4������������У��ָ������³�ѹʱ���������������ΪNA |
| A�� | �Ȼ��� | B�� | ���� | C�� | ��Ȳ | D�� | ���� |
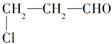 ����ȡ�۱�ϩ�ᣨ
����ȡ�۱�ϩ�ᣨ ���Ĺ����з����ķ�Ӧ����Ϊ��������
���Ĺ����з����ķ�Ӧ����Ϊ����������ȡ��������ȥ���ۼӾۡ��ܻ�ԭ����������
| A�� | �ݢ٢ڢ� | B�� | �ݢڢ� | C�� | �٢ڢݢ� | D�� | �ڢۢݢ� |
| A�� | �Ӻ�ˮ����ȡ�壬���������������������Һ���������ˮ��Һ��Ϊ����Һ | |
| B�� | �Ӻ�ˮ����ȡ�ⳣ�ù�����������ԭ�� | |
| C�� | ���������ȷ�Ӧ�Ƶõ���þ | |
| D�� | ���ȼ�����ָ��������������� |
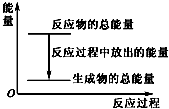
| A�� | ��Ӧ�����е�������ϵ������ͼ��ʾ | |
| B�� | 1 mol Zn����������1 mol H2������ | |
| C�� | ���������Ϊԭ��أ���п������ | |
| D�� | ���������Ϊԭ��أ�����32.5 g Zn�ܽ�ʱ�������ų�������һ��Ϊ11.2 L |
| A�� | c��N2����c��H2����c��NH3��=1��3��2 | |
| B�� | N2��H2��NH3�������������ٸı� | |
| C�� | N2��H2�����ʵ���֮����NH3�����ʵ�����2�� | |
| D�� | ��λʱ����ÿ����1 mol N2��ͬʱ����2 mol NH3 |
| A�� | �����ƺ�ʯ���Լ� | B�� | Na2CO3��Һ��ʯ���Լ� | ||
| C�� | ����KMnO4����ˮ | D�� | NaOH��Һ��CuSO4��Һ |
| A�� | Fe | B�� | Ag | C�� | Mg | D�� | Na |