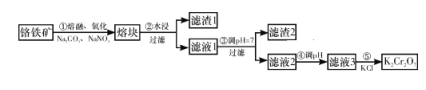
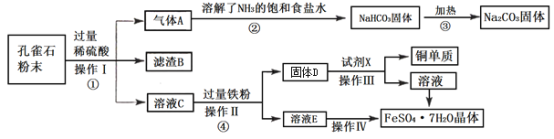
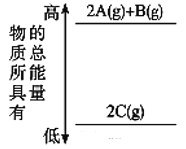
��Ŀ����
����Ŀ��������������Ҫ�Ĵ�����Ⱦ��֮һ����ҵ����һ����������NH3�ɽ�NO2��ԭ���ĺ�ͬѧ��ʵ���ҶԸ÷�Ӧ������̽����ʵ���������(���ּг�װ��ʡ��):
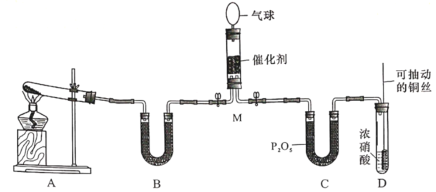
�ش���������:
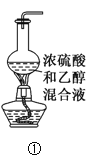
��1����װ��A�Ʊ�NH3,���з�����Ӧ�Ļ�ѧ����ʽΪ________________;װ��B�ڵ��Լ���______________
��2��װ��D�з�����Ӧ�����ӷ���ʽ��_________________��ͭ˿�ɳ鶯���ŵ���________________��
��3��NH3��NO2��M�г�ַ�Ӧ�������ֶԻ����Ѻõ����ʣ��÷�Ӧ�Ļ�ѧ����ʽΪ________________��M�пɹ۲쵽��������________________��
���𰸡�2NH4Cl��Ca(OH)2![]() CaCl2��2NH3����2H2O ��ʯ�ң���NaOH���壬��CaO���壩 Cu��4H+��2
CaCl2��2NH3����2H2O ��ʯ�ң���NaOH���壬��CaO���壩 Cu��4H+��2![]() =Cu2+��2NO2����2H2O ���ڿ��Ʒ�Ӧ�Ŀ�ʼ��ֹͣ 8NH3��6NO2
=Cu2+��2NO2����2H2O ���ڿ��Ʒ�Ӧ�Ŀ�ʼ��ֹͣ 8NH3��6NO2 7N2��12H2O ����ɫ��dz�������ܱ���Һ��
7N2��12H2O ����ɫ��dz�������ܱ���Һ��
��������
��1��װ��A�ǹ̹̼����͵�װ�ã��Ʊ�NH3��Ӧѡ���Ȼ�狀��������Ƽ��ȷ�Ӧ�������Ȼ��ƣ�ˮ�Ȱ�������Ӧ�Ļ�ѧ����ʽΪ2NH4Cl��Ca(OH)2![]() CaCl2��2NH3����2H2O��װ��B�������Ǹ��ﰱ������ѡ����Ը���������ʯ�ң���NaOH���壬��CaO���壩���ʱ����Ϊ��2NH4Cl��Ca(OH)2
CaCl2��2NH3����2H2O��װ��B�������Ǹ��ﰱ������ѡ����Ը���������ʯ�ң���NaOH���壬��CaO���壩���ʱ����Ϊ��2NH4Cl��Ca(OH)2![]() CaCl2��2NH3����2H2O����ʯ�ң���NaOH���壬��CaO���壩��
CaCl2��2NH3����2H2O����ʯ�ң���NaOH���壬��CaO���壩��
��2��װ��D��ͭ��Ũ���ᷢ����Ӧ����������ͭ������������ˮ�����ӷ���ʽ��Cu��4H+��2![]() =Cu2+��2NO2����2H2O��ͭ˿���³鶯��ʹ��Ӧ����룬�ŵ��DZ��ڿ��Ʒ�Ӧ�Ŀ�ʼ��ֹͣ���ʱ����Ϊ��Cu��4H+��2
=Cu2+��2NO2����2H2O��ͭ˿���³鶯��ʹ��Ӧ����룬�ŵ��DZ��ڿ��Ʒ�Ӧ�Ŀ�ʼ��ֹͣ���ʱ����Ϊ��Cu��4H+��2![]() =Cu2+��2NO2����2H2O�����ڿ��Ʒ�Ӧ�Ŀ�ʼ��ֹͣ��
=Cu2+��2NO2����2H2O�����ڿ��Ʒ�Ӧ�Ŀ�ʼ��ֹͣ��
��3��NH3��NO2��M�г�ַ�Ӧ�����ɵ�����ˮ���÷�Ӧ�Ļ�ѧ����ʽΪ8NH3��6NO2 7N2��12H2O��M�ж������������ģ�Ũ�Ƚ��ͣ��ɹ۲쵽�������Ǻ���ɫ��dz�������ܱ���Һ�Σ��ʱ����Ϊ��8NH3��6NO2
7N2��12H2O��M�ж������������ģ�Ũ�Ƚ��ͣ��ɹ۲쵽�������Ǻ���ɫ��dz�������ܱ���Һ�Σ��ʱ����Ϊ��8NH3��6NO2 7N2��12H2O������ɫ��dz�������ܱ���Һ�Ρ�
7N2��12H2O������ɫ��dz�������ܱ���Һ�Ρ�