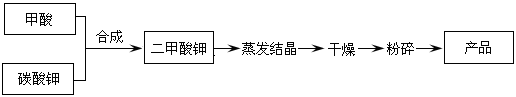
��Ŀ����
��֪���ᡢ��ˮ���ܶ�������ˮ���Ĺ�ϵ��ͼ��ʾ�����������백ˮ��һ�ݣ�����ݱ�����Ϣ���ش��������⣺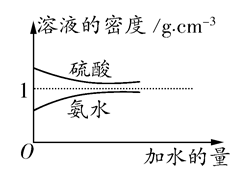
| | ���ʵ����ʵ��� Ũ��(mol��L��1) | ��Һ���ܶ�(g��cm��3) |
| ���� | c1 | ��1 |
| ��ˮ | c2 | ��2 |
(1)�����������������Ϊ (��д��λ���ú�c1����1�Ĵ���ʽ��ʾ)��
(2)���ʵ���Ũ��Ϊc1 mol��L��1��������ˮ��������(��Ϻ���Һ����仯���Բ���)��������Һ�����ʵ���Ũ��Ϊ mol��L��1��
(3)���ʵ���Ũ��Ϊc2 mol��L��1�İ�ˮ��
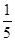 c2 mol��L��1�İ�ˮ��������ϣ�������Һ���ܶ� (����ڡ�����С�ڡ����ڡ�����ͬ)��2 g��cm��3��������Һ�����ʵ���Ũ��
c2 mol��L��1�İ�ˮ��������ϣ�������Һ���ܶ� (����ڡ�����С�ڡ����ڡ�����ͬ)��2 g��cm��3��������Һ�����ʵ���Ũ�� 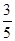 c2 mol��L��1(���Ϻ���Һ������仯���Բ���)��
c2 mol��L��1(���Ϻ���Һ������仯���Բ���)��
(1)  % (2)c1/2 (3)���ڴ���
% (2)c1/2 (3)���ڴ���
����

 53������ϵ�д�
53������ϵ�д�NA���������ӵ�����������˵����ȷ���ǣ� ��
| A����״���£�22.4 LSO3�к��еķ�����ΪNA |
| B��100 mL 2.0 mol/L�������������Һ�������Ӿ�Ϊ0.2NA |
| C��0.1mol/Lϡ�����к�SO42-����ĿΪ0.1NA |
| D����״���£�11.2LNO��5.6LO2��Ϻ�,��������С��0.5NA |
���ǻ��õļ����Ԫ�أ��Ƽ��仯�������������������й㷺��Ӧ�á�
������м��㣺
(1)��������(NaN3)��ײ����ȫ�ֽ�����ƺ͵������ʿ�Ӧ����������ȫ���ҡ�������40.32 L(��״����)������������Ҫ��������________g��
(2)���غϽ���ں˷�Ӧ���������Ƚ���Һ��5.05 g���غϽ�����200 mLˮ����0.075 mol������
�ټ�����Һ�����������ӵ����ʵ���Ũ��(������Һ����仯)��
_____________________________________________________________
�ڼ��㲢ȷ�������غϽ�Ļ�ѧʽ��
_____________________________________________________________
(3)����������Һ�����������ˣ��õ��������Ƶ���Һ�������Һ��ͨ�������̼�������з�Ӧ��
2NaAlO2��3H2O��CO2=2Al(OH)3����Na2CO3
��֪ͨ�������̼336 L(��״����)������24 mol Al(OH)3��15 mol Na2CO3����ͨ����Һ�Ķ�����̼Ϊ112 L(��״����)���������ɵ�Al(OH)3��Na2CO3�����ʵ���֮�ȡ�
_________________________________________________________________
(4)�����£���ȡ��ͬ����������Ʒ����ˮ���������к���pH��7��Ȼ����Һ���ɵ��Ȼ��ƾ��壬���ɹ����в�Ʒ����ʧ��
| | ������������(g) | �Ȼ�������(g) |
| �� | 2.40 | 3.51 |
| �� | 2.32 | 2.34 |
| �� | 3.48 | 3.51 |
����ʵ��٢ڢ������������ƾ��������ʣ���ʵ�����ݿɿ���ͨ�����㣬�����ͱȽ��ϱ�3�����ݣ��������ۡ�
 2O3��
2O3��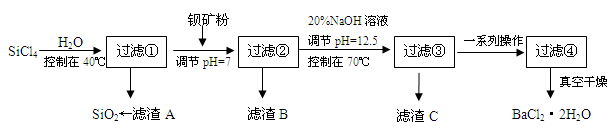
 ��Һ��
��Һ�� ��Һ�����ԣ�������ƽ��ԭ���ͱ�Ҫ�����ֽ�������C������3mol/L��
��Һ�����ԣ�������ƽ��ԭ���ͱ�Ҫ�����ֽ�������C������3mol/L��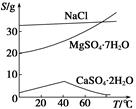
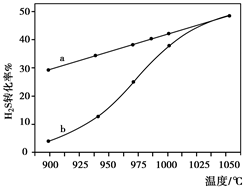
 H2(g)��1/2S2(g) �ں����ܱ������У����Ʋ�ͬ�¶Ƚ���H2S�ķֽ�ʵ�飺��H2S����ʼŨ�Ⱦ�Ϊc mol��L��1�ⶨH2S��ת���ʣ����������ͼ��ʾ��ͼ��aΪH2S��ƽ��ת�������¶ȹ�ϵ���ߣ�b���߱�ʾ��ͬ�¶��·�Ӧ������ͬʱ����δ�ﵽ��ѧƽ��ʱH2S��ת���ʡ���985��ʱ����Ӧ��t min�ﵽƽ�⣬��ʱH2S��ת����Ϊ40%����Ӧ����v(H2)�� ���ú�c��t�Ĵ���ʽ��ʾ������˵�����¶ȵ����ߣ�����b������a�ƽ���ԭ�� ��
H2(g)��1/2S2(g) �ں����ܱ������У����Ʋ�ͬ�¶Ƚ���H2S�ķֽ�ʵ�飺��H2S����ʼŨ�Ⱦ�Ϊc mol��L��1�ⶨH2S��ת���ʣ����������ͼ��ʾ��ͼ��aΪH2S��ƽ��ת�������¶ȹ�ϵ���ߣ�b���߱�ʾ��ͬ�¶��·�Ӧ������ͬʱ����δ�ﵽ��ѧƽ��ʱH2S��ת���ʡ���985��ʱ����Ӧ��t min�ﵽƽ�⣬��ʱH2S��ת����Ϊ40%����Ӧ����v(H2)�� ���ú�c��t�Ĵ���ʽ��ʾ������˵�����¶ȵ����ߣ�����b������a�ƽ���ԭ�� ��