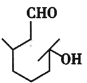
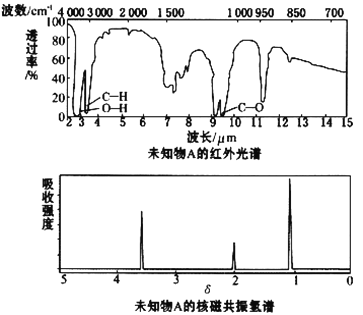
��Ŀ����
����Ŀ��ij�о�С���û�����(FeS2)�������ƺ�������Һ��Ϸ�Ӧ�Ʊ�ClO2���壬����ˮ���ո�����ɵ�ClO2��Һ���ڴ˹�������Ҫ�������˵��¶ȣ����¶Ȳ���������Ӧ���ӣ�Ӱ������ClO2����Ĵ��ȣ��һ�Ӱ��ClO2�������ʣ����������ͼ��ʾ��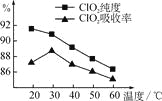
��1�� ��ͼ��֪����Ӧʱ��Ҫ���Ƶ������¶���________����Ҫ�ﵽ��Ҫ����Ҫ��ȡ�Ĵ�ʩ��______________��
��2�� ��֪���������е���Ԫ�������������¿ɱ�ClO3-������SO42-����д��FeS2�������ƺ�������Һ��Ϸ�Ӧ���ɶ�������(ClO2)�����ӷ���ʽ��______________________��
��3�� ��С��������![]() ����Ϊ����ClO2���ʵ�ָ�ꡣ��ȡNaClO3��Ʒ6.0 g��ͨ����Ӧ�����ջ��400 mL ClO2��Һ��ȡ����Һ20 mL��37.00 mL 0.500 mol��L��1(NH4)2Fe(SO4)2��Һ��ַ�Ӧ������Fe2������0.050 0 mol��L��1K2Cr2O7����Һ�ζ����յ㣬����K2Cr2O7����Һ20.00 mL����Ӧԭ��Ϊ��4H����ClO2��5Fe2��=Cl����5Fe3����2H2O��14H����Cr2O72-��6Fe2��=2Cr3����6Fe3����7H2O��
����Ϊ����ClO2���ʵ�ָ�ꡣ��ȡNaClO3��Ʒ6.0 g��ͨ����Ӧ�����ջ��400 mL ClO2��Һ��ȡ����Һ20 mL��37.00 mL 0.500 mol��L��1(NH4)2Fe(SO4)2��Һ��ַ�Ӧ������Fe2������0.050 0 mol��L��1K2Cr2O7����Һ�ζ����յ㣬����K2Cr2O7����Һ20.00 mL����Ӧԭ��Ϊ��4H����ClO2��5Fe2��=Cl����5Fe3����2H2O��14H����Cr2O72-��6Fe2��=2Cr3����6Fe3����7H2O��
�Լ���ClO2���������� _______________ (д���������)��
���𰸡�30�� ˮԡ���ȣ���ˮԡ���ƺ��£� 15ClO3-+FeS2+14H+=15ClO2+Fe3++7H2O+2SO42- ��K2Cr2O7��Ӧ��n��Fe2+��=6n��Cr2O72-��=6��20��10-3L��0.0500mol/L=6��10-3mol����ClO2��Ӧ��n��Fe2+��=37��10-3L��0.0500mol/L-6��10-3mol=1.25��10-2mol��n(ClO2)=![]() ��1.25��10-2mol=2.5��10-3mol��ClO2�IJ���=
��1.25��10-2mol=2.5��10-3mol��ClO2�IJ���=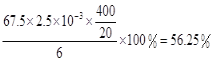
��������
��1������ͼ������¶���30��CʱClO2�����������90%���ϣ�����100��C���µ� �¶���Ҫ��ˮԡ���ȣ�
��2���û�����FeS2���������ƺ�������Һ��Ϸ�Ӧ�Ʊ�ClO2���壬����ԭ���غ㣬�����غ�͵���غ���ƽ��д���ӷ���ʽ��
��3�����ݻ�ѧ����ʽ�Ķ�����ϵ��ʽ���㣬�ԡ�![]() ����Ϊ����ClO2���ʵ�ָ�ꡣ
����Ϊ����ClO2���ʵ�ָ�ꡣ
��1����ͼ��֪����20�淴ӦʱClO2������ȡ���ȸߣ���������������ʽϵͣ���30�������Ƿ�Ӧ��ȡClO2���崿�ȸ�������������ʶ��ϸߡ����¶������ߣ���ȡClO2���崿�ȸ�������������ʶ����͡�������Ҫ���Ƶ������¶���30�棻�ﵽ��Ҫ���ȡ�����˴�ʩ��ˮԡ���ȣ���ˮԡ���ƺ��£���
(2)�������е���Ԫ�������������±�ClO3��������SO42�����Ʊ��������ȵ����ӷ���ʽΪ15ClO3����FeS2��14H��=15ClO2����Fe3����7H2O��2SO42����
��3���ɷ���ʽ4H++ClO2+5Fe2+=Cl-+5Fe3+ +2H2O��14H+ + Cr2O72-+6 Fe2+ =2Cr3+ + 6 Fe3+ +7H2O�ɽ�ϵ��ӵ�ʧ�غ�ɵ�1��n(Fe2+)=5��n(ClO2)+2��3��n(Cr2O72-)����K2Cr2O7��Ӧ��n��Fe2+��=6n��Cr2O72-��=6��20��10-3L��0.0500mol/L=6��10-3mol����ClO2��Ӧ��n��Fe2+��=37��10-3L�� 0.0500mol/L -6��10-3mol=1.25��10-2mol������n(ClO2)=![]() ��1.25��10-2mol=2.5��10-3mol��ClO2�IJ���=
��1.25��10-2mol=2.5��10-3mol��ClO2�IJ���= ��
��

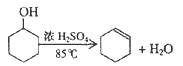
����Ŀ��ij��ѧС���������������������װ�ã���ͼ1)���Ի������Ʊ�����ϩ
��֪:

�ܶ�(g/cm3) | �۵㣨�棩 | �е㣨�棩 | �ܽ��� | |
������ | 0.96 | 25 | 161 | ������ˮ |
����ϩ | 0.81 | -103 | 83 | ������ˮ |
��1���Ʊ���Ʒ
��12.5mL�����������Թ�A�У��ټ���lmLŨ���ᣬҡ�Ⱥ�������Ƭ������������Ӧ��ȫ�����Թ�C�ڵõ�����ϩ��Ʒ��
��A�����Ƭ��������____________������B���˵�������е�������____________��
���Թ�C���ڱ�ˮԡ�е�Ŀ����______________________��
��2���Ʊ���Ʒ
�ٻ���ϩ��Ʒ�к��л������������������ʵȡ����뱥��ʳ��ˮ�������á��ֲ㣬����ϩ��______�㣨���ϻ��£�����Һ����___________(�����ţ�ϴ�ӡ�
a.KMnO4��Һ b.ϡH2SO4 c.Na2CO3��Һ
���ٽ�����ϩ��ͼ2��ʾװ��������ȴˮ��________�ڽ��룬�ռ���Ʒʱ�����Ƶ��¶�Ӧ��______���ҡ�
��3���������ֻ���ϩ��Ʒ����Ʒ�ķ�������������_____________��
a.�����Ը��������Һ b.��NaOH��Һ c.�ⶨ�е�