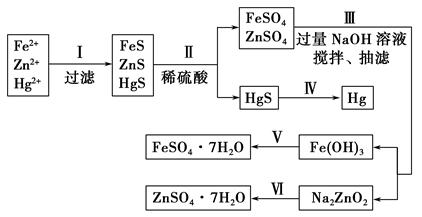
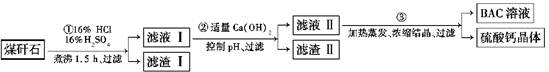
��Ŀ����
�������������������м�����Ҫ�����ã��������й��ա���Ч���Ͷ���ɱ�������������ǽ��չ�����ơ�
(1)Cl2��H2O2��ClO2(��ԭ����ΪCl��)��O3(1 mol O3ת��Ϊ1 mol O2��1 mol H2O)�����ʳ��������������������ʵ�����������������Ч����ߵ���________(�����)��
| A��Cl2 | B��H2O2 | C��ClO2 | D��O3 |
(3)Ư����������(NaClO2)�ڳ�����ڰ����ɱ���һ�ꡣ������ȶ��ɷֽ⣬��Ӧ�����ӷ���ʽΪHClO2�D��ClO2����H����Cl����H2O(δ��ƽ)���ڸ÷�Ӧ�У�����1 mol ClO2����ʱת�Ƶĵ��Ӹ���ԼΪ________��
(4)��84������Һ(��Ҫ�ɷ���NaClO)�ͽ��(��Ҫ�ɷ���Ũ����)���ܻ��ã�ԭ����__________________(�����ӷ���ʽ��ʾ)�������ȼҵ�IJ������������84������Һ��д���йط�Ӧ�Ļ�ѧ����ʽ��____________��
(1)C
(2)KHCO3��H2O2�����������������H2O��û����Ⱦ
(3)6.02��1023
(4)ClO����Cl����2H��=Cl2����H2O��Cl2��2NaOH=NaClO��NaCl��H2O
����

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д����ӷ�Ӧ����ѧ��ѧ����Ҫ�ķ�Ӧ���͡��ش��������⣺
(1)�ڷ������ӷ�Ӧ�ķ�Ӧ����������У�һ�������� (�����)��
�ٵ��ʣ���������۵���ʣ����Σ��ݻ�����
(2)����ͼʾ�ķ�����ʾ��ͬ��Ӧ����֮��Ĺ�ϵ����ֽⷴӦ��������ԭ��Ӧ�ɱ�ʾΪ��ͼ����������ķ����л������ӷ�Ӧ���û���Ӧ��������ԭ��Ӧ����֮��Ĺ�ϵ��

(3)���ӷ���ʽ����Ҫ�Ļ�ѧ����������й����ӷ���ʽ��һЩ����۵㣬�������б���������Ӧ�ġ����ӷ���ʽ������Щ�۵㡣
| �����е����ӷ���ʽ�����Ա�ʾһ�෴Ӧ | |
| ������кͷ�Ӧ���ɱ�ʾΪH����OH��=H2O | |
| �����ӷ���ʽ�з����������ᡢ��ξ�Ҫ�ꡰ�������� | |
(4)���оٳ����ֲ�ͬ��������(�ᡢ���)֮��ķ�Ӧ�����Ƕ�Ӧ�����ӷ���ʽ�����á�Ba2����SO42��=BaSO4��������ʾ����д���йط�Ӧ�Ļ�ѧ����ʽ(3��)��
�� ��
�� ��
�� ��
��һ�ְ�ɫ��ĩ���������������Ӻ��������еļ��֡�
�����ӣ�S2����Cl����NO�� ��
�� ��
�� ��
�� ��
��
�����ӣ�Na����Mg2����Al3����Ba2����Fe2����Fe3����Cu2���� ��
��
���ð�ɫ��ĩ��������ʵ�飬�۲쵽���������£�
| ʵ����� | ���� |
| a.ȡ������ĩ����ˮ���� | ȫ���ܽ⡢ |
| ��Һ��ɫ�� | |
| b.��������Һ�����������������Һ�������� | ���������� |
| c.ȡ������ĩ�������� | ���������� |
| d.ȡ������ĩ����ϡH2SO4��ϡHNO3�Ļ��Һ | �а�ɫ�������� |
(1)��aʵ���У����ƶϷ�ĩ�в������� (�����ӷ��ţ���ͬ)��
(2)��bʵ���У����ƶϷ�ĩ�в������� ��
(3)��cʵ���У����ƶϷ�ĩ�в������� ��
(4)��dʵ���У����ƶϷ�ĩ�в������� ��һ������ ��
(5)���ϸ�ʵ������ȷ���Ƿ���ڵ������� ��
 ��Fe2����Fe3����CO
��Fe2����Fe3����CO ��SO
��SO ��Cl����NO
��Cl����NO �е�һ�ֻ��֣�ȡ����Һ��������ʵ�飬ʵ��������£�
�е�һ�ֻ��֣�ȡ����Һ��������ʵ�飬ʵ��������£�

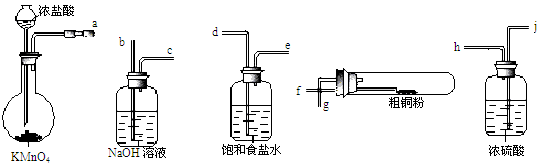
 CuCl42-(aq) + 4 H2O (l)
CuCl42-(aq) + 4 H2O (l)