��Ŀ����
ijʵ������480 mL 0.2mol/L��ϡ������Һ�������ܶ�Ϊ1.84g/mL����������Ϊ98%Ũ�������ƣ�����д���пհף�
��1����Ҫʹ�õ���Ҫ��������Ͳ���ձ����������� �� ��
��2�����������ɷֽ�Ϊ���¼�����
A������Ͳ��ȡ mLŨ���ᣬ����ע��װ��Լ50mL����ˮ���ձ��
���ò��������衣
B������������ˮ������ϴ���ձ��Ͳ���������ÿ�ε�ϴҺ����������ƿ�
C����ϡ�ͺ������С�ĵ��ò�������������ƿ�
D���������ƿ�Ƿ�©ˮ��
E��������ˮֱ�Ӽ�������ƿ����Һ��ӽ��̶���1-2cm����
F���ǽ�ƿ���������ߵ���ҡ����Һ��
G���ý�ͷ�ι�������ƿ����μ�������ˮ����Һ����͵�ǡ����������С�
��ݴ���д��
��������������еĿհ״���
�ڲ��������ȷ�IJ���˳������ĸ��д����
�� D ������ A ������ C ������ ������ ������ ������ F ����
�۽�Ũ�������ձ���ϡ�ͺ���������ƿʱ������_________________������Һ.
��3���Է������в�����������Һ��Ũ���к�Ӱ��
����ȡŨ����ʱ�۾����ӿ̶��ߣ��ᵼ��������ҺŨ�Ȼ� ����� ƫ�ߡ�ƫ�͡�����Ӱ�죬��ͬ��
�ڶ���ʱ���۾����ӿ̶��ߣ�������ҺŨ�Ȼ� ��
�۶��ݺӸǵ�תҡ�Ⱥ���������ڿ̶��ߣ��ֵμ�����ˮ���̶ȡ�������ҺŨ�Ȼ� .
��1����Ҫʹ�õ���Ҫ��������Ͳ���ձ����������� �� ��
��2�����������ɷֽ�Ϊ���¼�����
A������Ͳ��ȡ mLŨ���ᣬ����ע��װ��Լ50mL����ˮ���ձ��
���ò��������衣
B������������ˮ������ϴ���ձ��Ͳ���������ÿ�ε�ϴҺ����������ƿ�
C����ϡ�ͺ������С�ĵ��ò�������������ƿ�
D���������ƿ�Ƿ�©ˮ��
E��������ˮֱ�Ӽ�������ƿ����Һ��ӽ��̶���1-2cm����
F���ǽ�ƿ���������ߵ���ҡ����Һ��
G���ý�ͷ�ι�������ƿ����μ�������ˮ����Һ����͵�ǡ����������С�
��ݴ���д��
��������������еĿհ״���
�ڲ��������ȷ�IJ���˳������ĸ��д����
�� D ������ A ������ C ������ ������ ������ ������ F ����
�۽�Ũ�������ձ���ϡ�ͺ���������ƿʱ������_________________������Һ.
��3���Է������в�����������Һ��Ũ���к�Ӱ��
����ȡŨ����ʱ�۾����ӿ̶��ߣ��ᵼ��������ҺŨ�Ȼ� ����� ƫ�ߡ�ƫ�͡�����Ӱ�죬��ͬ��
�ڶ���ʱ���۾����ӿ̶��ߣ�������ҺŨ�Ȼ� ��
�۶��ݺӸǵ�תҡ�Ⱥ���������ڿ̶��ߣ��ֵμ�����ˮ���̶ȡ�������ҺŨ�Ȼ� .
����ÿ��1��,��8�֣�(1)500 ml����ƿ ��ͷ�ι�
(2)��5.4 ��B��E��G ����ȴ������ (3)��ƫ�� ��ƫ�� ��ƫ��
(2)��5.4 ��B��E��G ����ȴ������ (3)��ƫ�� ��ƫ�� ��ƫ��
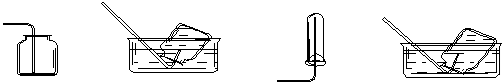
�����������1������ʵ����û��480ml������ƿ������Ӧ������500ml��Һ�������Ҫ����������500ml����ƿ�Ͷ���ʱ�Ľ�ͷ�ιܡ�
��2������ҪŨ����������
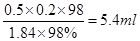 ��
����ת������������ϴ���ձ��Ͳ���������ϴ��ҺҲҪת�Ƶ�����ƿ�У�������ȷ��˳���ǣ� D ������ A ������ C ������B ������E ������ G ������ F ����
�۽�Ũ�������ձ���ϡ�ͺ���������ƿʱ��������ȴ�����·�����Һ��
��3������n��c��V��֪����ȡŨ����ʱ�۾����ӿ̶��ߣ��������������٣���������ҺŨ�Ȼ�ƫ�ͣ�����ʱ���۾����ӿ̶��ߣ�������ƿ����Һ��������٣���Ũ��ƫ�ߣ����ݺӸǵ�תҡ�Ⱥ���������ڿ̶��ߣ��ֵμ�����ˮ���̶ȣ�����Һ��������ӣ�������ҺŨ�Ȼ�ƫ�͡�
�������������е��Ѷȵ����⣬���������ǿ�������߿��������������У�ע������ԣ����ض�ѧ�������������ͽ��ⷽ����ָ����ѵ��������������ѧ������˼ά�������Ͻ��Ĺ淶ʵ�����������������ѵ���������������Ҫ��ȷ���Ǹ���cB��nB/V�ɵã�һ�����ʵ���Ũ����Һ���Ƶ����������ʵ����ʵ�����B����Һ�����V����ġ�������ʱ���ؼ�Ҫ�����ƹ�������������V�����ı仯��������һ�����ʵ���Ũ����Һʱ����nB������ֵС����V������ֵ��ʱ������ʹ������ҺŨ��ƫС����nB������ֵ��V������ֵСʱ������ʹ������ҺŨ��ƫ��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
 R-Br+H2O ��
R-Br+H2O ��