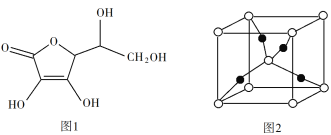
ЬтФПФкШн
ЁОЬтФПЁПГЃЮТЯТЃЌгаЯТСаЮхжжШмвКЃК
Ђй | Ђк | Ђл | Ђм | Ђн |
0.1 molЁЄL-1 CH3COOHШмвК | 0.01molЁЄL-1 CH3COOHШмвК | pHЃН2 CH3COOHШмвК | 0.1 molЁЄL-1 NaOHШмвК | 0.1molЁЄL-1 АБЫЎ |
ЛиД№ЯТСаЮЪЬтЃК
ЃЈ1ЃЉШмвКЂйЯЁЪЭЕНдРДЕФ10БЖКѓЕФШмвКpH______ЂлШмвКЕФpHЃЈЬюЁАЃОЁБЛђЁАЃНЁБЛђЁАЃМЁБЃЌЯТЭЌЃЉЃЌЂйКЭЂмСНШмвКжаЫЎЕчРыГіЕФc(HЃЋ)ЃКЂй_______Ђм
ЃЈ2ЃЉдкЯрЭЌЮТЖШЪБ100mL ЂкЕФШмвКгы10mL ЂйЕФШмвКЯрБШНЯЃЌЯТСаЪ§жЕЧАепДѓгкКѓепЕФЪЧ_______
A жаКЭЪБЫљашNaOHЕФСП B ЕчРыГЬЖШ
C ЫЎЕчРыГіЕФc(HЃЋ) D CH3COOHЕФЮяжЪЕФСП
ЃЈ3ЃЉгУЫЎЯЁЪЭЂнЪБЃЌШмвКжаЫцзХЫЎСПЕФдіМгЖјМѕаЁЕФЪЧ______ЃЈЬюзжФИЃЉЁЃ
AЁЂ ЁЁЁЁЁЁЁЁЁЁЁЁЁЁ BЁЂ
ЁЁЁЁЁЁЁЁЁЁЁЁЁЁ BЁЂ
CЁЂcЃЈH+ЃЉКЭcЃЈOH-ЃЉЕФГЫЛ§ЁЁЁЁЁЁЁЁDЁЂ OH-ЕФЮяжЪЕФСП
ЃЈ4ЃЉСЊАБЃЈN2H4ЃЉШмгкЫЎЯдМюадЃЌЦфдРэгыАБЯрЫЦЃЌЕЋЦфМюадВЛШчАБЧПЃЌаДГіЦфШмгкЫЎГЪМюадЕФРызгЗНГЬЪНЃК________ЁЃ
ЁОД№АИЁПЃО ЃО BC B N2H4+H2O![]() N2H5+ +OH-
N2H5+ +OH-
ЁОНтЮіЁП
ЃЈ1ЃЉЂйЕФХЈЖШЮЊ0.1mol/LЃЌpH=2ЕФCH3COOHШмвКЕФХЈЖШДѓгк0.01mol/LЃЌЯЁЪЭЕНдРДЕФ10БЖЃЌЂлЕФЧтРызгХЈЖШДѓЃЛЂйКЭЂмСНШмвКЖдЫЎЕФЕчРыЦ№вжжЦзїгУЃЌЧтРызгЛђЧтбѕИљРызгХЈЖШдНДѓЖдЫЎЕФЕчРыЕФвжжЦГЬЖШдНДѓЃЌЫЎЕчРыГіЕФcЃЈH+ЃЉдНаЁЃЛ
ЃЈ2ЃЉдкЯрЭЌЮТЖШЪБЃЌ100mLЂкЕФШмвКгы10mLЂйЕФШмвКЯрБШНЯЃЌ
AЁЂСНШмвКжаДзЫсЕФЮяжЪЕФСПЯрЭЌЃЌжаКЭЪБЫљашNaOHЕФСПЯрЕШЃЛ
BЁЂдНЯЁдНЕчРыЃЛ
CЁЂКѓепШмвКжаЧтРызгХЈЖШДѓгкЧАепЃЌЫљвдЫЎЕчРыГіЕФcЃЈH+ЃЉЧАепДѓгкКѓепЃЛ
DЁЂCH3COOHЕФЮяжЪЕФСПЃЌвђЮЊЧАепЕФЕчРыГЬЖШДѓгкКѓепЃЌЫљвдCH3COOHЕФЮяжЪЕФСПЧАепаЁгкКѓепЃЛ
ЃЈ3ЃЉМгЫЎДйНјАБЫЎЕФЕчРыЃЌnЃЈOH-ЃЉБфДѓЃЌnЃЈNH3H2OЃЉБфаЁЃЌдђ
AЁЂ ЯрЕБгк
ЯрЕБгк БфДѓЃЛ
БфДѓЃЛ
BЁЂ ЯрЕБ
ЯрЕБ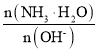 гкМѕаЁЃЛ
гкМѕаЁЃЛ
CЁЂЮТЖШВЛБфЃЌСНепcЃЈH+ЃЉКЭcЃЈOH-ЃЉЕФГЫЛ§ВЛБфЃЛ
DЁЂOH-ЕФЮяжЪЕФСПБфДѓЃЛ
ЃЈ4ЃЉЛЏКЯЮяЕФгІгУЃЌАБЦјгыЫЎЕФЗДгІЕФаджЪЃЈСЊАБЪЧвЛжжЮоЩЋПЩШМЕФвКЬхЃЌШмгкЫЎЯдМюадЃЌЦфдРэгыАБЯрЫЦЃЉЃЛРћгУ NH3+H2O![]() NH4++OH-ПЩвдЗжЮіГіСЊАБгыЫЎЗДгІЕФЗНГЬЪНЁЃ
NH4++OH-ПЩвдЗжЮіГіСЊАБгыЫЎЗДгІЕФЗНГЬЪНЁЃ
ЃЈ1ЃЉЂйCH3COOHШмвКЕФХЈЖШЮЊ0.1mol/LЃЌЯЁЪЭЕНдРДЕФ10БЖЃЌХЈЖШБфЮЊ0.01 molЁЄL-1ЃЌгЩгкДзЫсЪЧШѕЫсЃЌВЛЭъШЋЕчРыЃЌЫљвдЦфPHЃО2ЁЃ0.1 molЁЄL-1CH3COOHШмвКжаЕФc(H+)аЁгк0.1 molЁЄL-1NaOHШмвКжаЕФc(OH-)ЃЌЫљвдЫЎЕчРыГіЧтРызгЕФХЈЖШЂйЃОЂкЃЛЙЪД№АИЮЊЃКЃОЃЌЃОЃЛ
ЃЈ2ЃЉдкЯрЭЌЮТЖШЪБЃЌ100mL Ђк0.01molЁЄL-1 CH3COOHШмвКгы10mL Ђй0.1 molЁЄL-1CH3COOHШмвКЯрБШНЯЃЌ
AЁЂСНШмвКжаШмжЪЕФЮяжЪЕФСПЯрЭЌЃЌжаКЭЪБЫљашNaOHЕФСПЯрЕШЃЌЙЪВЛбЁЃЛ
BЁЂдНЯЁдНЕчРыЃЌЕчРыГЬЖШдНДѓЃЌЙЪбЁЃЛ
CЁЂКѓепШмвКжаЧтРызгХЈЖШДѓгкЧАепЃЌЫљвдЫЎЕчРыГіЕФcЃЈH+ЃЉЧАепДѓгкКѓепЃЌЙЪбЁЃЛ
DЁЂCH3COOHЕФЮяжЪЕФСПЃЌвђЮЊЧАепЕФЕчРыГЬЖШДѓгкКѓепЃЌЫљвдCH3COOHЕФЮяжЪЕФСПЧАепаЁгкКѓепЃЌЙЪВЛбЁЃЛ
ЙЪбЁBCЃЛ
ЃЈ3ЃЉAЁЂ ЯрЕБгк
ЯрЕБгк БфДѓЃЛ
БфДѓЃЛ
BЁЂ ЯрЕБгк
ЯрЕБгк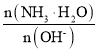 МѕаЁЃЛ
МѕаЁЃЛ
CЁЂЮТЖШВЛБфЃЌСНепcЃЈH+ЃЉКЭcЃЈOH-ЃЉЕФГЫЛ§ВЛБфЃЛ
DЁЂOH-ЕФЮяжЪЕФСПБфДѓЃЛЙЪбЁBЃЛ
ЃЈ4ЃЉСЊАБШмгкЫЎЯдМюадЃЌЦфдРэгыАБЯрЫЦМДN2H4+H2O![]() NH2NH3++OH-ЃЌНјвЛВНЗДгІвВПЩвдЮЊNH2NH3++H2O
NH2NH3++OH-ЃЌНјвЛВНЗДгІвВПЩвдЮЊNH2NH3++H2O![]() NH3NH32++OH-ЁЃ
NH3NH32++OH-ЁЃ

ЁОЬтФПЁПIЃЎФГбЇЩњгУ0.100molL-1ЕФKOHБъзМШмвКЕЮЖЈЮДжЊХЈЖШЕФбЮЫсЃЌЦфВйзїПЩЗжНтЮЊШчЯТМИВНЃК
AЃЎгУБъзМШмвКШѓЯДЕЮЖЈЙм2ЁЋ3ДЮЃЛ
BЃЎШЁБъзМKOHШмвКзЂШыМюЪНЕЮЖЈЙмжСПЬЖШЁА0ЁБвдЩЯ2ЁЋ3cm ДІЃЛ
CЃЎАбЪЂгаБъзМШмвКЕФМюЪНЕЮЖЈЙмЙЬЖЈКУЃЌЕїНкЕЮЖЈЙмЪЙМтзьДІГфТњШмвКЃЛ
DЃЎЕїНквКУцжСЁА0ЁБЛђЁА0ЁБвдЯТПЬЖШЃЌМЧЯТЖСЪ§ЃЛ
EЃЎвЦШЁ20mLД§ВтбЮЫсШмвКзЂШыНрОЛЕФзЖаЮЦПжаЃЌВЂМгШы2ЁЋ3ЕЮЗгЬЊЃЛ
FЃЎАбзЖаЮЦПЗХдкЕЮЖЈЙмЕФЯТУцЃЌгУБъзМKOHШмвКЕЮЖЈжСжеЕуВЂМЧЯТЕЮЖЈЙмвКУцЕФПЬЖШЁЃ
ЪЕбщБрКХ | KOHШмвКЕФХЈЖШ(mol/LЃЉ | ЕЮЖЈЭъГЩЪБЃЌKOHШмвКЕЮШыЕФЬхЛ§ЃЈmLЃЉ | Д§ВтбЮЫсШмвКЕФЬхЛ§ЃЈmLЃЉ |
1 | 0.10 | 22.62 | 20.00 |
2 | 0.10 | 22.72 | 20.00 |
3 | 0.10 | 22.80 | 20.00 |
ОЭДЫЪЕбщЭъГЩЬюПеЃКИљОнЩЯЪіЪ§ОнЃЌПЩМЦЫуГіИУбЮЫсЕФХЈЖШдМЮЊ__________ЃЈБЃСєСНЮЛгааЇЪ§зжЃЉ
IIЃЎбЧСђЫсбЮЪЧвЛжжГЃМћЪГЦЗЬэМгМСЁЃЮЊМьбщФГЪГЦЗжабЧСђЫсбЮКЌСП(ЭЈГЃвд1kgбљЦЗжаКЌSO2ЕФжЪСПМЦ)ЃЌФГбаОПаЁзщгУЁАЕтбѕЛЏЛЙдЕЮЖЈЗЈЁБНјааВтЖЈЃЌЪЕбщСїГЬШчЯТЃК

ЃЈ1ЃЉЕтБъзМвКгІбЁгУ____(ЬюЁАЫсЪНЁБЛђЁАМюЪНЁБ)ЕЮЖЈЙмЪЂзАЃЌМгзЂБъзМвКжЎЧАБиаыЖдЕЮЖЈЙмНјаа_____ЁЂЯДЕгЁЂ____ЁЃ
ЃЈ2ЃЉзЖаЮЦПФкЕФжИЪОМСЮЊ_____ЃЌХаЖЯДяЕНЕЮЖЈжеЕуЕФЯжЯѓЪЧ______________________ЁЃ
ЃЈ3ЃЉЯТСаВйзїЛсЪЙВтЖЈНсЙћЦЋДѓЕФга_______
AЃЎЦ№ЪМЖСЪ§ЪБЦНЪгЃЌжеЕуЖСЪ§ЪБИЉЪг
BЃЎЮДгУБъзМвКШѓЯДЕЮЖЈЙм
CЃЎВНжшЂйжагУЯЁбЮЫсДњЬцЯЁСђЫс
ЃЈ4ЃЉШєШЁбљЦЗw gЃЌАДввЗНАИВтЕУЯћКФ0.01000molЁЄL-1 I2ШмвКVmLЃЌдђ1kgбљЦЗжаКЌSO2ЕФжЪСПЪЧ____g(гУКЌwЁЂVЕФДњЪ§ЪНБэЪО)ЁЃ
ЁОЬтФПЁПЖўбѕЛЏСђЪЧЮЃКІзюЮЊбЯжиЕФДѓЦјЮлШОЮяжЎвЛ,ЫќжївЊРДздЛЏЪЏШМСЯЕФШМЩе,баОПCOДпЛЏЛЙдSO2ЕФЪЪвЫЬѕМў,дкШМУКЕчГЇЕФбЬЦјЭбСђжаОпгаживЊМлжЕЁЃ
Ђё.ДгШШСІбЇНЧЖШбаОПЗДгІ
(1) C(s)+O2(g)![]() CO2(g) ІЄH1=-393.5 kJЁЄmol-1
CO2(g) ІЄH1=-393.5 kJЁЄmol-1
CO2(g)+C(s)![]() 2CO(g) ІЄH2=+172.5 kJЁЄmol-1
2CO(g) ІЄH2=+172.5 kJЁЄmol-1
S(s)+O2(g)![]() SO2(g) ІЄH3=-296.0 kJЁЄmol-1
SO2(g) ІЄH3=-296.0 kJЁЄmol-1
аДГіCO ЛЙдSO2ЕФШШЛЏбЇЗНГЬЪН:_________________ЁЃ
(2)ЙигкCOЛЙдSO2ЕФЗДгІ,ЯТСаЫЕЗЈе§ШЗЕФЪЧ______ЁЃ
A.дкКуЮТКуШнЬѕМўЯТ,ШєЗДгІЬхЯЕбЙЧПВЛБф,дђЗДгІвбДяЕНЦНКтзДЬЌ
B.ЦНКтзДЬЌЪБ,2vе§(CO)=vФц(SO2)
C.ЦфЫћЬѕМўВЛБф,діДѓSO2ЕФХЈЖШ,COЕФЦНКтзЊЛЏТЪдіДѓ
D.дкКуЮТКубЙЕФШнЦїжа,ЯђДяЕНЦНКтзДЬЌЕФЬхЯЕжаГфШыN2,SO2ЕФЦНКтзЊЛЏТЪВЛБф
ЂђЃЎNOxЕФХХЗХжївЊРДздгкЦћГЕЮВЦјЃЌАќКЌNO2КЭNOЃЌгаШЫЬсГігУЛюадЬПЖдNOxНјааЮќИНЃЌЗЂЩњЗДгІШчЯТЃК
ЗДгІaЃКC(s)+2NO(g)![]() N2(g)+CO2(g) ІЄH=Ѓ34.0kJ/mol
N2(g)+CO2(g) ІЄH=Ѓ34.0kJ/mol
ЗДгІbЃК2C(s)+2NO2(g)![]() N2(g)+2CO2(g) ІЄH=Ѓ64.2kJ/mol
N2(g)+2CO2(g) ІЄH=Ѓ64.2kJ/mol
(3)ЖдгкЗДгІaЃЌдкT1ЁцЪБЃЌНшжњДЋИаЦїВтЕУЗДгІдкВЛЭЌЪБМфЕуЩЯИїЮяжЪЕФХЈЖШШчЯТЃК
ЪБМф(min) ХЈЖШ(molЁЄL-1) | 0 | 10 | 20 | 30 | 40 | 50 |
NO | 1.00 | 0.58 | 0.40 | 0.40 | 0.48 | 0.48 |
N2 | 0 | 0.21 | 0.30 | 0.30 | 0.36 | 0.36 |
Ђй0~10minФкЃЌNOЕФЦНОљЗДгІЫйТЪv(NO)=___________ЃЌЕБЩ§ИпЗДгІЮТЖШЃЌИУЗДгІЕФЦНКтГЃЪ§K___________(бЁЬюЁАдіДѓЁБЁЂЁАМѕаЁЁБЛђЁАВЛБфЁБ)ЁЃ
Ђк30minКѓЃЌжЛИФБфФГвЛЬѕМўЃЌЗДгІжиаТДяЕНЦНКтЃЛИљОнЩЯБэжаЕФЪ§ОнХаЖЯИФБфЕФЬѕМўПЩФмЪЧ___________(ЬюзжФИ)ЁЃ
AЃЎМгШывЛЖЈСПЕФЛюадЬП BЃЎЭЈШывЛЖЈСПЕФNO
CЃЎЪЪЕБЫѕаЁШнЦїЕФЬхЛ§ DЃЎМгШыКЯЪЪЕФДпЛЏМС
(4)ЂйФГЪЕбщЪвФЃФтЗДгІbЃЌдкУмБеШнЦїжаМгШызуСПЕФCКЭвЛЖЈСПЕФNO2ЦјЬхЃЌЮЌГжЮТЖШЮЊT2ЁцЃЌШчЭМЮЊВЛЭЌбЙЧПЯТЗДгІbОЙ§ЯрЭЌЪБМфNO2ЕФзЊЛЏТЪЫцзХбЙЧПБфЛЏЕФЪОвтЭМЁЃЧыДгЖЏСІбЇНЧЖШЗжЮіЃЌ1050kPaЧАЃЌЗДгІbжаNO2зЊЛЏТЪЫцзХбЙЧПдіДѓЖјдіДѓЕФдвђ_____________ЃЛдк1100kPaЪБЃЌNO2ЕФЬхЛ§ЗжЪ§ЮЊ___________ЁЃ

ЂкгУФГЮяжЪЕФЦНКтЗжбЙДњЬцЦфЮяжЪЕФСПХЈЖШвВПЩвдБэЪОЛЏбЇЦНКтГЃЪ§(МЧзїKp)ЃЛдкT2ЁцЁЂ1.1ЁС106PaЪБЃЌИУЗДгІЕФЛЏбЇЦНКтГЃЪ§Kp=___________(МЦЫуБэДяЪНБэЪО)ЃЛвбжЊЃКЦјЬхЗжбЙ(PЗж)=ЦјЬхзмбЙ(Pзм)ЁСЬхЛ§ЗжЪ§ЁЃ