��Ŀ����
���ֶ�����Ԫ��A��B��C��D��E��ԭ��������������A��Cͬ�壬B��Dͬ�壬C���Ӻ�B���Ӿ�����ͬ�ĵ��Ӳ�ṹ��A��B��D��E�����γɹ��ۻ����A��B�γɵĻ�������ˮ�гʼ��ԣ�C��E�γɵĻ�������ˮ�г����ԡ�
�ش��������⣺
��1������Ԫ���У�ԭ�Ӱ뾶������ ���ǽ�������ǿ���� (��Ԫ�ط���)��
��2����A��B��D��E���γɵĹ����ͻ������У����ȶ��������� ���û�ѧʽ��ʾ����
��3��A��E�γɵĻ�������A��B�γɵĻ����ﷴӦ������Ļ�ѧʽΪ ,���д��ڵĻ�ѧ������Ϊ ��
��4������D�ڳ���ĵ���E��ȼ�գ���Ӧ�Ļ�ѧ����ʽΪ ��
��5������E��ˮ��Ӧ�����ӷ���ʽ ��
��1��Na Cl
��2��PH3
��3��NH4Cl ���Ӽ������ۼ�
��4��2P+5Cl2 2PCl5
2PCl5
��5��Cl2+H2O=H++ Cl-+HClO
�������������A��B�γɵĻ�������ˮ�гʼ��ԣ��ж�A��HԪ�أ�B��NԪ�أ�A��Cͬ�壬B��Dͬ�壬��C��NaԪ�أ�D��PԪ�أ�C��E�γɵĻ�������ˮ�г����ԣ���E��ClԪ�أ��Ȼ��Ƶ���Һ�����ԡ�
��1������Ԫ���������жϣ�����Ԫ���У�ԭ�Ӱ뾶������Na���ǽ�������ǿ����Cl��
��2��B��D��E�зǽ�������������P����H�γ�PH3�����ȶ�����
��3��A��E�γɵĻ�������HCl��A��B�γɵĻ������ǰ��������߷�Ӧ�����Ȼ�炙�ѧʽΪNH4Cl���������Ӽ������ۼ���
��4��P��������ȼ���������Ȼ��ף���ѧ����ʽΪ2P+5Cl2 2PCl5
2PCl5
��5��������ˮ��Ӧ�����Ȼ���ʹ����ᣬ���ӷ���ʽΪCl2+H2O=H++ Cl-+HClO
���㣺����Ԫ���ƶϣ�Ԫ�������ɵ�Ӧ�ã���ѧ�����жϣ���ѧ����ʽ�����ӷ���ʽ����д

 Ӧ������ҵ��ϵ�д�
Ӧ������ҵ��ϵ�д��±�ΪԪ�����ڱ���һ���֣���ش��й����⣺
| | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | | | | | �� | �� | �� | |
| 3 | �� | �� | �� | �� | �� | �� | �� | |
��1����������õĽ����� ���ǽ�������ǿ��Ԫ���� ����дԪ�ط��ţ���
��2���������γ��������������Ԫ�������� ��д����Ԫ�ص�����������ܵ�����������Ӧˮ���ﷴӦ�����ӷ���ʽ�� ��
��3��Ԫ�آ��Ԫ�آ������������ˮ���������Խ�ǿ�� ���û�ѧʽ��ʾ����
��4��Ԫ�آ���Ԫ�آ��γɵĻ��������� (�� �����ۡ������ӡ�)�����
��5���뻭��Ԫ�آڵ�ԭ�ӽṹʾ��ͼΪ ��Ԫ�آٵĵ����ڳ����»�ѧ�����ȶ���ͨ�������������������õ���ʽ��ʾ����� ��
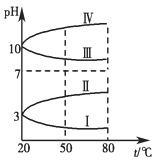
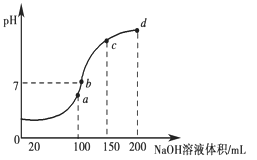
 ����֪��Ԫ�ص�ԭ�Ӻ�������������������ȣ����Ԫ�ص�������________��
����֪��Ԫ�ص�ԭ�Ӻ�������������������ȣ����Ԫ�ص�������________��