��Ŀ����
18������˵����ȷ���ǣ�������| A�� | �����ȼ����Ϊ 890.3 kJ•mol-1�������ȼ�յ��Ȼ�ѧ����ʽ�ɱ�ʾΪCH4��g��+2O2��g���TCO2��g��+2H2O��g����H=-890.3 kJ•mol | |
| B�� | 500�桢30 MPa �£��� 0.5 mol N2 �� 1.5 mol H2 �����ܱյ������г�ַ�Ӧ���� NH3��g�������� 19.3 kJ�����Ȼ�ѧ����ʽΪ N2��g��+3H2��g��?2NH2��g����H=-38.6 kJ•mol | |
| C�� | һ�������£���Ӧ N2��g��+3H2��g��?2NH3��g����H=-38.6 kJ•mol�����¶Ƚ�������Ӧ�����ƶ� | |
| D�� | ��֪ 2C��s��+2O2��g���T2CO2��g����H12C��s��+O2��g���T2CO��g����H2�����H1����H2 |
���� A��ȼ����ָ����1mol��ȼ����ȫȼ�������ȶ���������ʱ�ų���������ע�����ɵ�ˮӦΪҺ̬��
B��0.5molN2��1.5molH2�����ܱ������г�ַ�Ӧ����NH3��g��������19.3kJ�����ɵİ��������ʵ���С��1mol����ͬ�����µ�ͬһ���淴Ӧ�����淴Ӧ��Ӧ����ֵ��ȣ������෴��
C����Ӧ�Ƿ��ȷ�Ӧ�����¶ȣ�ƽ��������У�
D��һ����̼ȼ�����ɶ�����̼�����Ƿ��ȷ�Ӧ��
��� �⣺A��ȼ����ָ����1mol��ȼ����ȫȼ�������ȶ���������ʱ�ų���������ע�����ɵ�ˮӦΪҺ̬������Ϊ��̬����A����
B����ͬ�����µ�ͬһ���淴Ӧ�����淴Ӧ��Ӧ����ֵ��ȣ������෴��0.5molN2��1.5molH2�����ܱ������г�ַ�Ӧ����NH3��g��������19.3kJ�����ɵİ��������ʵ���С��1mol������2mol�����ֽ⣬���յ���������38.6kJ����B����
C��һ�������£���Ӧ N2��g��+3H2��g��?2NH3��g����H=-38.6 kJ•mol����Ӧ�Ƿ��ȷ�Ӧ�������¶Ƚ�������Ӧ�����ƶ�����C��ȷ��
D��һ����̼ȼ�����ɶ�����̼�����Ƿ��ȷ�Ӧ���ʱ�Ϊ��ֵ�����H1����H2����D����
��ѡC��
���� ���⿼�����Ȼ�ѧ����ʽ��д����Ӧ�ȱȽϣ�Ӱ�컯ѧƽ������ط����жϣ����ջ����ǹؼ�����Ŀ�ϼ�

| A�� | ����ʯ��ˮ������������Cl2+OH-�TCl-+ClO-+H+ | |
| B�� | NaHSO4��Һ��Ba��OH��2��Һ��Ӧ�����ԣ�2H++SO42-+Ba2++2OH-�TBaSO4��+2H2O | |
| C�� | ��ϡ�����м����������ۣ�3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O | |
| D�� | ��̼�������Һ�м����������Լ���Һ��Ca2++2HCO3-+2OH-�TCaCO3��+CO32-+2H2O |
C��s��+CO2��g���T2CO��g������H2
2CO��g��+O2��g���T2CO2��g������H3
4Fe��s��+3O2��g���T2Fe2O3��s������H4
Fe2O3��s��+3CO��g���T2Fe��s��+3CO2��g������H5
���й���������Ӧ�ʱ���ж���ȷ���ǣ�������
| A�� | ��H1��0����H5��0 | B�� | ��H2��0����H4��0 | C�� | ��H1=��H2+��H3 | D�� | ��H3=��H4+��H5 |
| A�� | ����Ħ�����Ϊ22.4L•mol-1 | |
| B�� | 1molH2��������2g������ռ�������22.4L | |
| C�� | 1molˮ�к���2mol���1mol�� | |
| D�� | �ڱ�״���£�1mol�κ�������ռ�������ԼΪ22.4L |
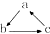 ��ϵ�ת�����ǣ�������
��ϵ�ת�����ǣ�������| ѡ�� | A | B | C | D |
| a | Fe | SiO2 | Al��OH��3 | HNO3 |
| b | FeCl3 | Na2SiO3 | NaAlO2 | NO |
| c | FeCl2 | H2SiO3 | Al2O3 | NO2 |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ֻ�л��ý�������÷ǽ���Ԫ��ԭ��֮������γ����Ӽ� | |
| B�� | Na2O2�������Ӻ������Ӹ�����Ϊ1��1 | |
| C�� | �γ����Ӽ������������Ӽ�ֻ�о��������� | |
| D�� | ���ۼ������ڹ��ۻ������У�Ҳ���ܴ����ڵ��ʼ����ӻ������� |
| A�� | ��������ͬ | B�� | ��ԭ�Ӹ������ | C�� | ��ԭ�Ӹ������ | D�� | ������� |
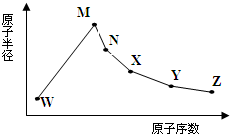 W��M��N��X��Y��Z�����ֳ����Ķ�����Ԫ�أ���ԭ�Ӱ뾶��ԭ�������仯��ͼ��ʾ����֪W����̬�⻯����ʹʪ��ĺ�ɫʯ����ֽ������M��Neԭ�ӵĺ�����������1��N��X���ڣ�X����������������ά����Ҫԭ�ϣ�Y�������������������ڲ��������3����Z�ķǽ�������ͬ��������Ԫ�������
W��M��N��X��Y��Z�����ֳ����Ķ�����Ԫ�أ���ԭ�Ӱ뾶��ԭ�������仯��ͼ��ʾ����֪W����̬�⻯����ʹʪ��ĺ�ɫʯ����ֽ������M��Neԭ�ӵĺ�����������1��N��X���ڣ�X����������������ά����Ҫԭ�ϣ�Y�������������������ڲ��������3����Z�ķǽ�������ͬ��������Ԫ�������