��Ŀ����
A��B��C��D��EΪԪ�����ڱ��ж�����Ԫ���γɵ��������ӣ�A��B��C��D�������ӵĵ���������E�ĵ�������8��
��1��A��B��ϳɵĻ�������м������Ӽ����й��ۼ���A��C��ϳɵĻ�������������ˮ�������ڼ�ˮ��Һ����ɫ����Һ��CԪ�������ڱ��е�λ��Ϊ _ �������ˮ��Һ��Ӧ�����ӷ���ʽΪ__________________________________��
��2��B��E��ϳɻ������������������֮��Ϊ2��1��A��D��ϳɻ����ﶡ���������Ӧ������ɫ��ζ�����壬����ĵ���ʽΪ�� _ ��1molD���������� _ mol����
��3����һ��Һ̬���⻯�����죬�������E�ĵ�������ͬ���ǡ������ߺš��ɴ�����ʱʹ�õĸ���ȼ��֮һ���ṹ�������ָ÷��ӽṹ��ֻ�е������백���ƣ�����ĽṹʽΪ ___��
��1����3���ڢ�A Al(OH)3+OH-=AlO2-+2H2O
��2�� 11mol
11mol
��3��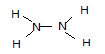
���������������1����������Ϊ10�����ӵ������ӣ�N3-��O2-��F-��OH-��NH2-�� �����ӣ�Na+��Mg2+��Al3+��NH4+��H3O+����������Ϊ18�����ӵ������ӣ�S2-��O22-��Cl-��HS-�� �����ӣ�K+��Ca2+��N2H5+��N2H62+�����������⣬A��B��C��D����10e-���ӣ�EΪ18e-���ӣ�A��B�γɵĻ�������м������Ӽ����й��ۼ���10e-�������γɵĻ�����������Ӽ����й��ۼ�������Ϊ���λ��������A��C��ϳɵĻ�������������ˮ��10e-�����н�ϳ�������ˮ�Ļ�������Mg��OH��2��Al��OH��3���ٸ��������ڼ�ˮ��Һ����ɫ����Һ��ȷ����ΪAl��OH��3����Ϊǿ��NaOH����AΪOH-��BΪNa+��CΪAl3+����C�����ڱ��е�λ�õ������ڣ���A�塣Al��OH��3��NaOH��Ӧ����ʽΪAl��OH��3+NaOH�TNaAlO2+2H2O����д���ӷ���ʽΪAl��OH��3+OH-=AlO2-+2H2O����2���������⣬A��OH-��D��H3O+����ϳɻ����ﶡ����˶�ΪH2O��B��Na+��B��E�γɵĻ������������������֮��Ϊ2��1��E����ΪS2-��O22-������Ϊ�������Ӧ������ɫ��ζ���壬���Ա�ֻ��ΪNa2O2������ʽ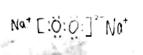 1mol H3O+��11mol���ӡ�
1mol H3O+��11mol���ӡ�
��3���������⣬��������ΪҺ̬���⻯���ͬʱҲΪ18���ӣ�Nԭ�Ӻ�����7�����ӣ�Hԭ�Ӻ�����1�����ӣ���˻���������Ӧ��2��Nԭ�Ӻ�4��Hԭ�ӣ��ʻ�ѧʽΪN2H4���ṹ�����õ��÷�����ֻ���е��������Ե�ԭ�Ӻ͵�ԭ�ӡ���ԭ�Ӻ���ԭ��֮�䶼���γ�һ�Թ��õ��Ӷԣ��ʵ���ʽΪ ���ṹʽ
���ṹʽ ��
��
���㣺���⿼��10���Ӻ�18������������ʽ���ṹʾ��ͼ�����ӷ���ʽ

 ��У����ϵ�д�
��У����ϵ�д������������(BN)n��һ�����ϳɲ��ϣ����ʽΪ(BN)�ݣ����и�Ӳ�ȡ����µ��ص㣬���������������մɲ��ϡ�ĥ�ϡ���ĥ�оߵĺò��ϡ�����ɰ��Na2B4O7���������ڸ��¸�ѹ�·�Ӧ���Ի�á����磺 Na2B4O7 + 2CO(NH2)2 �� 4(BN) + Na2O + 2CO2
��1���������������ʽ��ʾ��ɰ�Ļ�ѧʽ ������(BN)n�Ⱦ������и���Ӳ�Ⱥ������Ե�ԭ���ǣ� ��
��21��������Ӧʽ�о���4�ֲ�ͬ�������ӵ�ԭ�ӣ���������Ԫ�������ڱ��д��ڵ� ���ڣ��� �塣
��3�������ڱȽ�N��O�ǽ��������ǿ������ʵ�� ��
| A������������Ӧˮ��������� | B��H2O(g) ��NH3(g)�ȶ� |
| C��������H2��Ӧ�����׳̶� | D��NO�е�Ԫ�������ۣ���Ԫ���Ը��� |
��5������ͬ�������������ڵ�Ԫ�أ��������������NaOH��Һ��Ӧ�����ӷ���ʽ Ϊ�� ��

 HCNO + NH3������ƽ�������������գ�
HCNO + NH3������ƽ�������������գ� 2N2��g����3H2O��g��
2N2��g����3H2O��g��