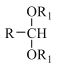��Ŀ����
����Ŀ��I-�ⶨ�Ļ���ԭ���ǽ���������I2������Na2S2O3����Һ���ζ��������Ʒ�н�������I-������������ѧ�Ŵ�����Ӧ����������Ŵ�����Ȼ���ٽ��вⶨ�������ǻ�ѧ�Ŵ�Ӧ��ʵ�鲽�裺
I����������I-����Ʒ��Һ�������Ի������ԣ�������ˮ����I-��ȫ������IO![]() �����ȥ���������壻
�����ȥ���������壻
��ȡ������Һ��������������ữ��KI��Һ������Һ��ɫ���ػ�ɫ��
����������Һ��ȫ����I2��ȡ��CCl4�У�
�������Һ���CCl4��Һ�м�����(N2H4)��ˮ��Һ����������N2����ȥ�л��㣻
V����������ˮ��Һ�ظ�I�����裻
�����Ե���Ϊָʾ������Na2S2O3����Һ�ζ����յ㡣��֪����ӦΪ��I2+2S2O![]() =2I-+S4O
=2I-+S4O![]() ��Na2S2O3��Na2S4O6��Һ��ɫ��Ϊ��ɫ��
��Na2S2O3��Na2S4O6��Һ��ɫ��Ϊ��ɫ��
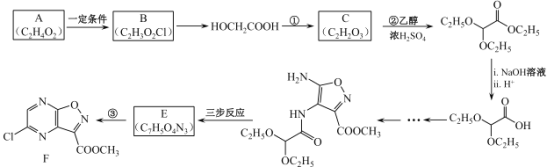
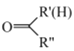
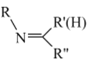
(1)I���������ͻ�ԭ�����ʵ���֮��Ϊ______��
(2)����ʹ�õ���Ҫ����������______(������)��
(3)���з�Ӧ�����ӷ���ʽ��______��
(4)������Ҫ�����Ի������Ի����н��У�pH����Na2S2O3�ױ�I2������Na2SO4�������ӷ���ʽΪ_______��������ӷ���ʽ����pH���˹�С��ԭ��______��VI�еζ��յ��������______��
(5)��Ʒ��I-���������ĵ�Na2S2O3�����ʵ�������Ʒ�г�ʼI-���ʵ����ı�ֵ��Ϊ���Ŵ��������������ϲ������Ŵ���Ϊ______��
���𰸡�3��1 ��Һ©�� N2H4+2I2=4I-+N2+4H+ S2O![]() +4I2+10OH-=2SO
+4I2+10OH-=2SO![]() +8I-+5H2O pH����S2O
+8I-+5H2O pH��С��S2O![]() +2H+=S+SO2+H2O��4I-+O2+4H+=2I2+2H2O ��Һ��ɫǡ����ʧ��30�벻�ָ� 36
+2H+=S+SO2+H2O��4I-+O2+4H+=2I2+2H2O ��Һ��ɫǡ����ʧ��30�벻�ָ� 36
��������
(1)I�н�������I-����Ʒ��Һ�������Ի������ԣ�������ˮ����I-��ȫ������IO![]() ��������Ϊ�壬��ԭ��ΪI-�����ݵ�ʧ�����غ�ϼ������غ�������㣻
��������Ϊ�壬��ԭ��ΪI-�����ݵ�ʧ�����غ�ϼ������غ�������㣻
(2)���ǽ���Һ�е�I2��ȡ��CCl4�У��ݴ˷������
(3)�������Һ���CCl4��Һ�м�����(N2H4)��ˮ��Һ����������N2���ǵ⽫�������Ĺ��̣��ݴ���д��Ӧ�����ӷ���ʽ��
(4)������Ҫ�����Ի������Ի����н��У�pH����Na2S2O3�ױ�I2������Na2SO4��pH��С��S2O![]() ��������Һ���ܹ������绯��Ӧ��������Һ�е��������ױ������е��������������ݷ�Ӧ��ԭ���жϵζ��յ������
��������Һ���ܹ������绯��Ӧ��������Һ�е��������ױ������е��������������ݷ�Ӧ��ԭ���жϵζ��յ������
(5)�����������裬�����ķ�ӦΪI-+3Br2+3H2O= IO![]() +6H++6Br-��IO
+6H++6Br-��IO![]() +5I-+6H+=3I2+3H2O��I2+2S2O
+5I-+6H+=3I2+3H2O��I2+2S2O![]() =2I-+S4O
=2I-+S4O![]() ���ڲ���V�н�������ˮ��Һ�ظ�I�����裬�ݴ˷����ж�I-���ʵ������Ŵ�ı�����
���ڲ���V�н�������ˮ��Һ�ظ�I�����裬�ݴ˷����ж�I-���ʵ������Ŵ�ı�����
(1)I�н�������I-����Ʒ��Һ�������Ի������ԣ�������ˮ����I-��ȫ������IO![]() ��������Ϊ�壬��ԭ��ΪI-�����ݵ�ʧ�����غ�ϼ������غ㣬�������ͻ�ԭ�������ʵ���֮��Ϊ
��������Ϊ�壬��ԭ��ΪI-�����ݵ�ʧ�����غ�ϼ������غ㣬�������ͻ�ԭ�������ʵ���֮��Ϊ![]() =
=![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��
(2)���ǽ���Һ�е�I2��ȡ��CCl4�У���ȡ�ͷ�Һ������ʹ�õ���Ҫ���������Ƿ�Һ©�����ʴ�Ϊ����Һ©����
(3)�������Һ���CCl4��Һ�м�����(N2H4)��ˮ��Һ����������N2���ǵ⽫�������Ĺ��̣���Ӧ�����ӷ���ʽ��N2H4+2I2=4I-+N2+4H+���ʴ�Ϊ��N2H4+2I2=4I-+N2+4H+��
(4)������Ҫ�����Ի������Ի����н��У�pH����Na2S2O3�ױ�I2������Na2SO4����Ӧ�����ӷ���ʽΪS2O![]() +4I2+10OH-=2SO
+4I2+10OH-=2SO![]() +8I-+5H2O��pH����S2O
+8I-+5H2O��pH��С��S2O![]() �ܹ������绯��Ӧ��S2O
�ܹ������绯��Ӧ��S2O![]() +2H+=S+SO2+H2O��������Һ�е��������ױ������е�����������4I-+O2+4H+=2I2+2H2O�����pHҲ���˹�С���Ե���Ϊָʾ������Na2S2O3����Һ�ζ�I2���յ㣬�ζ��յ������Ϊ��Һ��ɫǡ����ʧ��30�벻�ָ����ʴ�Ϊ��S2O
+2H+=S+SO2+H2O��������Һ�е��������ױ������е�����������4I-+O2+4H+=2I2+2H2O�����pHҲ���˹�С���Ե���Ϊָʾ������Na2S2O3����Һ�ζ�I2���յ㣬�ζ��յ������Ϊ��Һ��ɫǡ����ʧ��30�벻�ָ����ʴ�Ϊ��S2O![]() +4I2+10OH-=2SO
+4I2+10OH-=2SO![]() +8I-+5H2O��pH����S2O
+8I-+5H2O��pH��С��S2O![]() +2H+=S+SO2+H2O��4I-+O2+4H+=2I2+2H2O����Һ��ɫǡ����ʧ��30�벻�ָ���
+2H+=S+SO2+H2O��4I-+O2+4H+=2I2+2H2O����Һ��ɫǡ����ʧ��30�벻�ָ���
(5)�����������裬�����ķ�ӦΪI-+3Br2+3H2O= IO![]() +6H++6Br-��IO
+6H++6Br-��IO![]() +5I-+6H+=3I2+3H2O��I2+2S2O
+5I-+6H+=3I2+3H2O��I2+2S2O![]() =2I-+S4O
=2I-+S4O![]() ���ڲ���V�н�������ˮ��Һ�ظ�I�����裬��˵õ�I-��IO3-��3I2��6I-��6IO3-��18I2��36S2O32-����˾������ϲ�������ϵ�Ԫ���غ㣬��ʼI-���ʵ������Ŵ���36�����ʴ�Ϊ��36��
���ڲ���V�н�������ˮ��Һ�ظ�I�����裬��˵õ�I-��IO3-��3I2��6I-��6IO3-��18I2��36S2O32-����˾������ϲ�������ϵ�Ԫ���غ㣬��ʼI-���ʵ������Ŵ���36�����ʴ�Ϊ��36��

����Ŀ��ˮ������Ⱦ�ѳ�Ϊһ��ؽ�������ȫ���Ի������⣬�ҹ���ѧ���о��������������ƣ�Na2S2O8��ȥ����ˮ�е��������[As(��)]�������ģ����ͼ��
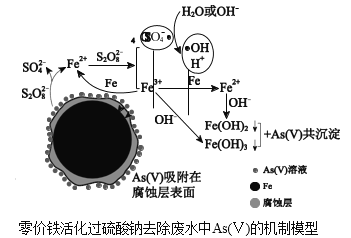
���ϣ�
��.����������SO![]() Ϊ��Ҫ�����ɻ������Լ�������������SO
Ϊ��Ҫ�����ɻ������Լ�������������SO![]() ������OHͬʱ���ڣ�ǿ������������OHΪ��Ҫ�����ɻ���
������OHͬʱ���ڣ�ǿ������������OHΪ��Ҫ�����ɻ���
��.Fe2+��Fe3+��������������pH
���� | ��ʼ������pH | ������ȫ��pH |
Fe2+ | 7.04 | 9.08 |
Fe3+ | 1.87 | 3.27 |
��1����������Ԫ�����ڱ���λ��ͬһ���壬��ԭ�ӱ���һ�����Ӳ㡣
������Ԫ�����ڱ��е�λ����_____��
������Ļ�ѧʽ��_____�������Ա�H3PO4_____������ǿ��������������
��2���������������Ʒ�Ӧ���ɳ����ͷ�Fe2+��Fe2+��S2O![]() ��Ӧ����Fe3+�����ɻ������ɻ�����ǿ�����ԣ������γ�Fe2+��Fe3+����ȷ��As(��)ȥ����ȫ��
��Ӧ����Fe3+�����ɻ������ɻ�����ǿ�����ԣ������γ�Fe2+��Fe3+����ȷ��As(��)ȥ����ȫ��
���������������Ʒ�Ӧ�����ӷ���ʽ��_____��
��Fe3+ת��ΪFe2+�����ӷ���ʽ��_____��
��SO![]() ����H2O��Ӧ�����ӷ���ʽ��_____��
����H2O��Ӧ�����ӷ���ʽ��_____��
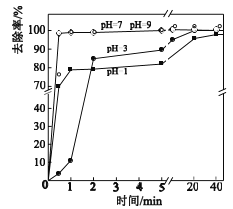
��3����ͬpH��As(��)ȥ���ʵ�Ӱ����ͼ��5min��pH=7��pH=9ʱȥ���ʸߵ�ԭ����_____��