��Ŀ����
�£�N2H4���ֳ���������һ�ֿ�ȼ��Һ�壬�������������ﷴӦ�������ɵ�����ˮ��������һ�������Դ��Һ����¾����������ȼ�ϡ�
�� ��������ȡ�봢��������Դ����������о��ȵ㡣
��֪��
�� ��������ȡ�봢��������Դ����������о��ȵ㡣
��֪��

��1��������Ϊ����Դ���ŵ�___________________������2�㣩
��2���Լ���Ϊԭ����ȡ�����ǹ�ҵ�ϳ��õ����ⷽ���� ��Ӧ����
��Ӧ����
 ���Ȼ�ѧ����ʽΪ___________________ ��
���Ȼ�ѧ����ʽΪ___________________ ��
��3��H2O���ȷֽ�Ҳ�ɵõ�H2��������ˮ�ֽ���ϵ����Ҫ���������������¶ȵĹ�ϵ��ͼ��ʾ��ͼ��A��B��ʾ������������__________��_____________��
��2���Լ���Ϊԭ����ȡ�����ǹ�ҵ�ϳ��õ����ⷽ����
 ��Ӧ����
��Ӧ���� ���Ȼ�ѧ����ʽΪ___________________ ��
���Ȼ�ѧ����ʽΪ___________________ ����3��H2O���ȷֽ�Ҳ�ɵõ�H2��������ˮ�ֽ���ϵ����Ҫ���������������¶ȵĹ�ϵ��ͼ��ʾ��ͼ��A��B��ʾ������������__________��_____________��

��4����--����ȼ�ϵ����һ�ּ���ȼ�ϵ�أ��������Һ��20%~30%��KOH��Һ���õ�طŵ�ʱ�������ĵ缫��Ӧʽ��_________________��
��5����ͼ��һ���绯ѧװ��ʾ��ͼ������--����ȼ�ϵ������װ�õĵ�Դ��
��5����ͼ��һ���绯ѧװ��ʾ��ͼ������--����ȼ�ϵ������װ�õĵ�Դ��

�����A�Dz��缫��B��ʯī�缫��C�����ᡪ����泥������ĵ缫��Ӧʽ��_________��
�����ø�װ�ÿ��Ƶ������������⣺��������SO42-��������S2O82-��������������ӣ���S2O82-��H2O��Ӧ����H2O2��S2O82-+2H2O=2SO42-+H2O2+2H+����Ҫ��ȡ2molH2O2����ȼ�ϵ��������������_______molN2H4��
��6����A��B��C��D���ֽ������±���װ�ý���ʵ�顣
ʵ��װ��������
�����ø�װ�ÿ��Ƶ������������⣺��������SO42-��������S2O82-��������������ӣ���S2O82-��H2O��Ӧ����H2O2��S2O82-+2H2O=2SO42-+H2O2+2H+����Ҫ��ȡ2molH2O2����ȼ�ϵ��������������_______molN2H4��
��6����A��B��C��D���ֽ������±���װ�ý���ʵ�顣
ʵ��װ��������

����ʵ������ش��������⣺
��װ�ñ�����Һ��pH______����������С�����䡱��
�����ֽ���������������ǿ��˳����_________________ ��
��װ�ñ�����Һ��pH______����������С�����䡱��
�����ֽ���������������ǿ��˳����_________________ ��
��1������Ⱦ����Դ�ḻ ����ֵ�ߵ�
��2��CH4(g)+2H2O(g)=CO2(g)+4H2(g) ��H=+165kJ/mol
��3����ԭ�ӣ���ԭ��
��4��N2H4-4e-+4OH-=N2+4H2O
��5����2H++2e-=H2����1
��6���ٱ��CBAD
��2��CH4(g)+2H2O(g)=CO2(g)+4H2(g) ��H=+165kJ/mol
��3����ԭ�ӣ���ԭ��
��4��N2H4-4e-+4OH-=N2+4H2O
��5����2H++2e-=H2����1
��6���ٱ��CBAD

��ϰ��ϵ�д�
 ��ĩ�����ϵ�д�
��ĩ�����ϵ�д�
�����Ŀ
 �£�N2H4���ֳ���������һ�ֿ�ȼ��Һ�壬�������������ﷴӦ�������ɵ�����ˮ���������ȼ�ϣ�
�£�N2H4���ֳ���������һ�ֿ�ȼ��Һ�壬�������������ﷴӦ�������ɵ�����ˮ���������ȼ�ϣ� �£�N2H4���ֳ��������㷺���ڻ���ƽ������л��ϳɼ�ȼ�ϵ�أ�NO2�Ķ�����N2O4���ǻ���г��õ�����������ش��������⣺
�£�N2H4���ֳ��������㷺���ڻ���ƽ������л��ϳɼ�ȼ�ϵ�أ�NO2�Ķ�����N2O4���ǻ���г��õ�����������ش��������⣺
 �£�N2H4���ֳ���������һ�ֿ�ȼ��Һ�壬�������ȼ�ϣ�
�£�N2H4���ֳ���������һ�ֿ�ȼ��Һ�壬�������ȼ�ϣ�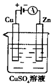 �£�N2H4���ֳ����������������ȼ�ϣ���һ����ȼ�ϵ���еĵ������Һ��20%��30%��KOH��Һ����ȼ�ϵ�ؿ���Ϊ��ͼװ���еĵ�Դ�������жϴ�����ǣ�������
�£�N2H4���ֳ����������������ȼ�ϣ���һ����ȼ�ϵ���еĵ������Һ��20%��30%��KOH��Һ����ȼ�ϵ�ؿ���Ϊ��ͼװ���еĵ�Դ�������жϴ�����ǣ�������