��Ŀ����
����Ŀ������֤�����淴ӦN2 + 3H22NH3�Ѵﵽƽ��״̬����( )
��һ��N��N���ѵ�ͬʱ����3��H��H������
��һ��N��N�����ѵ�ͬʱ����6��N��H������
��������������ʱ���������ƽ��ʽ�����ٸı�
�ܱ���������������ʱ����ϵѹǿ���ٸı�
��NH3����N2%��H2%�����ٸı�
���º���ʱ���ܶȱ��ֲ���
������Ӧ����v(H2)=0.6mol/(L��min)���淴Ӧ����v(NH3)=0.4mol/(L��min)
A.ȫ��B.ֻ�Тۢܢ�C.�ڢۢܢݢ�D.ֻ�Тۢݢޢ�
���𰸡�C
��������
��һ��N��N���ѵ�ͬʱ����3��H��H�����ѣ������Ķ�������Ӧ������˵����Ӧ�Ƿ�ƽ�⣬�ٴ���
��һ��N��N�����ѵ�ͬʱ����6��N��H�����ѣ�������Ӧ���ʺ��淴Ӧ������ȣ��Ѵ�ƽ�⣬����ȷ��
������Ӧ��һ�����������ʵ���n��С������������m����ķ�Ӧ����n=![]() ��֪�����������ƽ��ʽ�����ٸı䣬��M���䣬��֮����������m���䣬�����������ʵ���n���䣬˵����ƽ�⣬����ȷ��
��֪�����������ƽ��ʽ�����ٸı䣬��M���䣬��֮����������m���䣬�����������ʵ���n���䣬˵����ƽ�⣬����ȷ��
������Ӧ�Ǹ�����ѹǿ��С�ķ�Ӧ������ϵѹǿ���ٸı䣬˵����Ӧ�Ѵ�ƽ�⣬����ȷ��
��NH3����N2%��H2%�����ٸı䣬˵����ϵ�и����ʵĺ������ٸı䣬�Ѵ�ƽ�⣬����ȷ��
�÷�Ӧ�����������䣬������䣬�����۷�Ӧ�Ƿ�ƽ�⣬�ܶȶ����䣬���ܶȱ��ֲ��䣬����˵���÷�Ӧ�Ƿ�ƽ�⣬����
������Ӧ����v��(H2)=0.6mol/(L��min)����v��(NH3)=0.4mol/(L��min)������Ϊ�淴Ӧ����v��(NH3)=0.4mol/(L��min)�����ԣ�v��(NH3) =v��(NH3)��˵����Ӧ�Ѵ�ƽ�⣬����ȷ��
�����������ڢۢܢݢ�˵����Ӧ�Ѵ�ƽ�⣬C�������⡣
��ѡC��

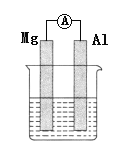
 �����������Ů��ͯ������ϵ�д�
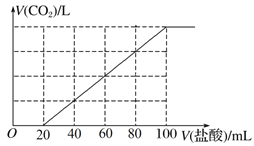
�����������Ů��ͯ������ϵ�д�����Ŀ��ijѧ��Ϊ��̽��п�����ᷴӦ�����е����ʱ仯������100 mLϡ�����м���������п�ۣ�����ˮ�������ռ���Ӧ�ų�������(��״��)��ʵ���¼����(�ۼ�ֵ)��
ʱ��/min | 1 | 2 | 3 | 4 | 5 |
�������/mL | 50 | 120 | 232 | 290 | 310 |
(1)��0��1 min��1��2 min��2��3 min��3��4 min��4��5 minʱ����У���Ӧ��������ʱ�����________��ԭ��Ϊ______________________����Ӧ������С��ʱ�����________��ԭ��Ϊ__________________________��
(2)��2��3 min�ڣ��������Ũ�ȱ仯��ʾ�ķ�Ӧ����Ϊ________��
(3)Ϊ�˼�����Ӧ���ʵ������ٲ��������������������зֱ����������������Һ�����п��е���________��
A������ˮ B��Na2SO4��Һ
C��NaNO3��Һ D��Na2CO3��Һ