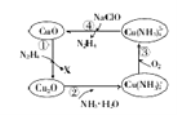
��Ŀ����
����Ŀ����Դ�ͻ������������������ע���ȵ㻰�⡣��ش��������⣺
����Ŀǰ����̼����������Ϊ��ѧ���о�����Ҫ���⡣��������������һ���Ƚ������ⷽ�����䷴Ӧ����ʽΪCH4(g) +H2O (g) =CO (g) +3H2(g) ��
�Ķ���ͼ������÷�Ӧ�ķ�Ӧ����H=____________kJ��mol-1��
�����ռ�������CO2�ǻ����������ȵ���⡣
500��ʱ�����ݻ�Ϊ1L���ܱ������г���1 mol CO2��3 mol H2���������·�Ӧ��CO2(g) +3H2 (g) ![]() CH3OH (g) +H2O (g) ��H��0�����CO2��CH3OH��Ũ����ʱ��Ĺ�ϵ��ͼ��ʾ��
CH3OH (g) +H2O (g) ��H��0�����CO2��CH3OH��Ũ����ʱ��Ĺ�ϵ��ͼ��ʾ��

��1��0~10 min��v(H2)=_____________��
A�㺬����_____________��
�÷�Ӧƽ�ⳣ������ʽK=____________��
��2����Ӧ��500���ﵽƽ��ı䷴Ӧ�¶�ΪT��CH3OH��Ũ����ÿ����0��030 mol/L������5 min�ִﵽ��ƽ�⡣T____________(����>������<������=��)500�����ж�������___________________��
��3���¶�ΪTʱ����Ӧ�ﵽƽ�����Ӧ�������ݻ�����һ����ƽ����____________(����������������)��Ӧ�����ƶ����ж�������____________��
�����绯ѧ������SO2��Ŀǰ�о����ȵ㡣
����˫��ˮ����SO2������SO2��Ⱦ�����װ����ͼ��ʾ��
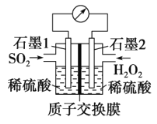
��1��ʯī1Ϊ___________(��������������������)��
�����ĵ缫��ӦʽΪ____________��
��2����11.2 L(��״��)SO2���뷴Ӧ����Ǩ��H+�����ʵ���Ϊ____________��
���𰸡�
����+161.1��
������1��0.225mol/��L�qmin������Ӧ3minʱ��c��CO2��= c��CH3OH����![]() ��
��
��2�������ı��¶ȣ�CH3OH��Ũ������ƽ��������Ӧ�����ƶ�����÷�Ӧ������ӦΪ���ȷ�Ӧ����T<500����
��3���棻�÷�Ӧ���淴Ӧ�������������ĵķ�Ӧ������������������൱�ڼ�Сѹǿ��ƽ���������������ķ����ƶ���
������1��������H2O2+2e-+2H+=2H2O����2��1mol��
��������
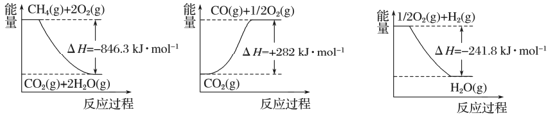
�������������������ͼ������CH4��g��+2O2��g����CO2��g��+2H2O��g����H=-846.3kJmoL-1����CO2��g���TCO��g��+![]() O2��g����H=+282kJmoL-1����
O2��g����H=+282kJmoL-1����![]() O2��g��+H2��g���TH2O��g����H=-241.8kJmoL-1����-�ۡ�3+����CH4��g��+H2O��g��
O2��g��+H2��g���TH2O��g����H=-241.8kJmoL-1����-�ۡ�3+����CH4��g��+H2O��g��![]() CO��g��+3H2��g����H=��-846.3+241.8��3+282��kJmoL-1=+161.1kJmoL-1���ʴ�Ϊ��+161.1��
CO��g��+3H2��g����H=��-846.3+241.8��3+282��kJmoL-1=+161.1kJmoL-1���ʴ�Ϊ��+161.1��
������1��Y����ʼŨ��Ϊ1mol/L����Y��ʾ������̼��ƽ��ʱ������̼��Ũ��Ϊ0.25mol/L����v��CO2��=![]() =0.075mol/��L�qmin��������֮�ȵ��ڻ�ѧ������֮�ȣ���v��H2��=3v��CO2��=3��0.075mol/��L�qmin��=0.225mol/��L�qmin��������ͼ��A���ʾ����Ӧ3minʱ��c��CO2��= c��CH3OH�����÷�Ӧƽ�ⳣ������ʽK=
=0.075mol/��L�qmin��������֮�ȵ��ڻ�ѧ������֮�ȣ���v��H2��=3v��CO2��=3��0.075mol/��L�qmin��=0.225mol/��L�qmin��������ͼ��A���ʾ����Ӧ3minʱ��c��CO2��= c��CH3OH�����÷�Ӧƽ�ⳣ������ʽK=![]() ���ʴ�Ϊ��0.225mol/��L�qmin������Ӧ3minʱ��c��CO2��= c��CH3OH����
���ʴ�Ϊ��0.225mol/��L�qmin������Ӧ3minʱ��c��CO2��= c��CH3OH����![]() ��
��
��2����Ӧ��500���ﵽƽ��ı䷴Ӧ�¶�ΪT��CH3OH��Ũ����ÿ����0��030 mol/L��������ƽ��������Ӧ�����ƶ�������CO2(g) +3H2 (g) ![]() CH3OH (g) +H2O (g) ��H��0��֪���¶��轵�ͣ��ʴ�Ϊ�������ı��¶ȣ�CH3OH��Ũ������ƽ��������Ӧ�����ƶ�����÷�Ӧ������ӦΪ���ȷ�Ӧ����T<500����
CH3OH (g) +H2O (g) ��H��0��֪���¶��轵�ͣ��ʴ�Ϊ�������ı��¶ȣ�CH3OH��Ũ������ƽ��������Ӧ�����ƶ�����÷�Ӧ������ӦΪ���ȷ�Ӧ����T<500����
��3���¶�ΪTʱ����Ӧ�ﵽƽ�����Ӧ�������ݻ�����һ����ѹǿ��С������CO2(g) +3H2 (g) ![]() CH3OH (g) +H2O (g) ��֪��ƽ�����淴Ӧ�����ƶ����ʴ�Ϊ���棻�÷�Ӧ���淴Ӧ�������������ĵķ�Ӧ������������������൱�ڼ�Сѹǿ��ƽ���������������ķ����ƶ���
CH3OH (g) +H2O (g) ��֪��ƽ�����淴Ӧ�����ƶ����ʴ�Ϊ���棻�÷�Ӧ���淴Ӧ�������������ĵķ�Ӧ������������������൱�ڼ�Сѹǿ��ƽ���������������ķ����ƶ���
������1������ͼʾ������������˫��ˮ����������ԭ��Ӧ�����ж��������ǻ�ԭ��������������Ӧ���������ᣬʯī1Ϊ������˫��ˮ����������������ԭ��Ӧ������ˮ��ʯī2���������缫��ӦΪH2O2+2e-+2H+=2H2O����Ϊ��������H2O2+2e-+2H+=2H2O��
��2��11.2 L(��״��)SO2�����ʵ���Ϊ0.5mol��ת�Ƶ���1mol������������Ӧʽ��Ǩ��H+�����ʵ���Ϊ1mol���ʴ�Ϊ��1mol��