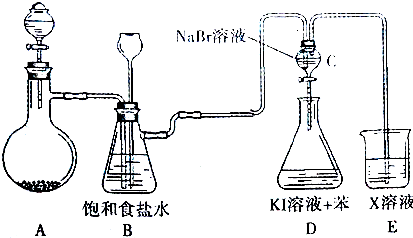
��Ŀ����
17����ͼ���ɶ�����Ԫ����ɵ�һЩ���ʼ��仯����֮���ת����ϵͼ���������ʾ�йص�һ�ַ�Ӧ��������ijЩ�����Ѿ���ȥ��������A��B��D�ڳ����¾�Ϊ��ɫ�̼�����ζ�����壬C��ʹʪ��ĺ�ɫʯ����ֽ���������壬M���������ɫҺ��
��1������G�Ļ�ѧʽΪHNO3������F�������Ƕ���������
��2����A��D�Ļ�ѧ����ʽΪ2CO2+2Na2O2=2Na2CO3+O2����C��E�Ļ�ѧ����ʽΪ4NH3+5O2$\frac{\underline{\;\;����\;\;}}{���¸�ѹ}$4NO+6H2O��
��3��G��E�����ӷ���ʽΪ3Cu+8H++2NO3-�T3Cu2++2NO��+4H2O��
���� �������и�����ת����ϵ��֪����ɫ��ζ����A��������Ʒ�Ӧ�ĵõ�����D����AΪCO2��DΪO2��C��ʹʪ��ĺ�ɫʯ����ֽ���������壬��CΪNH3����BΪN2��EΪNO��FΪNO2��M���������ɫҺ�壬ӦΪH2O����ת����ϵ��֪GΪHNO3���ݴ˽��н��
��� �⣺�������и�����ת����ϵ��֪����ɫ��ζ����A��������Ʒ�Ӧ�ĵõ�����D����AΪCO2��DΪO2��C��ʹʪ��ĺ�ɫʯ����ֽ���������壬��CΪNH3����BΪN2��EΪNO��FΪNO2��M���������ɫҺ�壬ӦΪH2O����ת����ϵ��֪GΪHNO3��
��1�����ݷ�����֪��GΪHNO3������FΪ����������
�ʴ�Ϊ��HNO3������������
��2����A��D�Ļ�ѧ����ʽΪ��2CO2+2Na2O2=2Na2CO3+O2��
�ʴ�Ϊ��2CO2+2Na2O2�T2Na2CO3+O2��
��C��E�Ļ�ѧ����ʽΪ��4NH3+5O2$\frac{\underline{\;\;����\;\;}}{���¸�ѹ}$4NO+6H2O��
�ʴ�Ϊ��4NH3+5O2$\frac{\underline{\;\;����\;\;}}{���¸�ѹ}$4NO+6H2O��
��3��G��EΪͭ��ϡ����ķ�Ӧ����Ӧ�����ӷ���ʽΪ��3Cu+8H++2NO3-�T3Cu2++2NO��+4H2O��
�ʴ�Ϊ��3Cu+8H++2NO3-�T3Cu2++2NO��+4H2O��
���� ���⿼��������ƶϣ���Ŀ�Ѷ��еȣ��漰N��NaԪ�ػ�����֪ʶ��������Ԫ�ػ�����֪ʶ�Ŀ��飬C��M�����ʼ�A����������Ʒ�Ӧ���ƶ�ͻ�ƿڣ�����������ѧ���ķ���������������������

 Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д� Сѧͬ�����������ܾ�ϵ�д�
Сѧͬ�����������ܾ�ϵ�д���Һ$��_{��}^{�ᴿ���ᾧ}$FeSO4•7H2O$\stackrel{��}{��}$FeSO4��Һ$��_{��}^{NH_{4}HCO_{3}��Һ}$FeCO3$\stackrel{��}{��}$Fe2O3
��1����֪����������������ڲ�ͬ�¶��µ��ܽ�Ⱥ����������������±���ʾ��
| �¶�/�� | 0 | 10 | 30 | 50 | 56.7 | 60 | 64 | 70 | 80 | 90 | ||
| �ܽ��/g | 14.0 | 17.0 | 25.0 | 33.0 | 35.2 | 35.3 | 35.6 | 33.0 | 30.5 | 27.0 | ||
| �������� | FeSO4•7H2O | FeSO4•4H2O | FeSO4•H2O | |||||||||
��2����������һ�������ᣬ����ҪĿ��������FeSO4��ˮ�⣮
��3������FeCO3��Һ�����ӷ���ʽΪFe2++2HCO3-�TFeCO3��+CO2��+H2O��FeCO3��Һ¶���ڿ����л���ֺ��ɫ�Ĺ��壬�ñ仯�Ļ�ѧ����ʽΪ4FeCO3+6H2O+O2�T4Fe��OH��3+4CO2��
��4����֪FeSO4•7H2O��Һ�����ڼ��������·������·�Ӧ��FeSO4•7H2O$\frac{\underline{\;����\;}}{\;}$Fe2O3+SO2��+SO3��+14H2O����������ͼװ�ÿɼ���÷�Ӧ��������
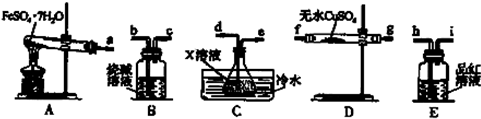
����д���пհף�
������������˳��Ϊa��f����g����g����f����d��e��h��i��b��
��װ��C�е�XΪ�Ȼ�����Һ��
| A�� | NH3��HCl | B�� | H2S��SO2 | C�� | NO��O2 | D�� | N2��O2 |
| A�� |  | B�� |  | ||
| C�� |  | D�� |  |
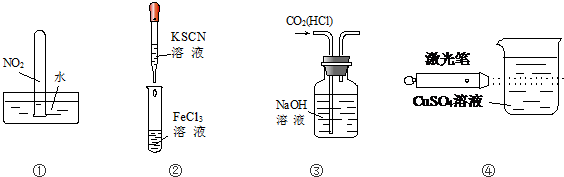
| A�� | ʵ����Թ����ջ����ˮ | |
| B�� | ʵ����Թ�����Һ��ΪѪ��ɫ | |
| C�� | ����ʵ���װ�ó�ȥCO2�����к��е�����HCl���� | |
| D�� | ʵ��ܿɹ۲춡������� |
| A�� | ��A�����A��Ԫ�ؼ���γɹ��ۻ���������ӻ����� | |
| B�� | �ڶ�����Ԫ�ش����ң�������۴�+1������+7 | |
| C�� | ͬ����Ԫ�صļ������ӻ�ԭ�Դ��ϵ������μ��� | |
| D�� | ͬ���ڽ���Ԫ�صĻ��ϼ�Խ�ߣ���ԭ��ʧ��������Խǿ |
| A�� | ��������ָ�����к���һ���������������� | |
| B�� | ����������ʹ���Ը��������Һ��ɫ | |
| C�� | ������������������Ӧ | |
| D�� | ������������ԭ�Ӷ���ͬһ��ƽ���� |
 ��F
��F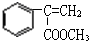
 ��
��