��Ŀ����
����Ŀ����ѧС���������ʵ�鷽�����ⶨij���ֱ���Ϊ̼���Ƶ�С�մ���Ʒ��NaHCO3������������
��1������һ:��ȡһ��������Ʒ����������a���þ����Ƽ�������������ȴ����������ƽ����ʣ������������ظ����������㡣
����a��������____________������������Ӧ�Ļ�ѧ����ʽΪ_______________��ʵ����Ϊ��֤NaHCO3�ֽ���ȫ���������������жϷ�����________________��
�Ʒ�����:��ȡһ��������Ʒ������С�ձ��У�������ˮ�ܽ�����С�ձ��м�������Ba��OH��2��Һ�����������ˣ�ϴ�ӣ���������������������㡣
д��NaHCO3������Ba��OH��2��Һ��Ӧ�����ӷ���ʽ_______________��ʵ�����жϳ����Ƿ���ȫ�ķ�����______________�����˲����У����ձ���©���⣬��Ҫ�õ��IJ�������Ϊ____________��
��3��������:��ȡһ��������Ʒ������ͼװ�ý���ʵ�飺
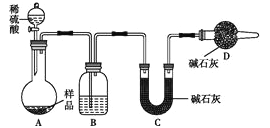
Bװ������ʢ�Լ�����Dװ�õ�������______________��ʵ��ǰ��ȡ17.9g��Ʒ��ʵ�����Cװ������8.8g������Ʒ��NaHCO3����������Ϊ________��ijͬѧ��Ϊ�����ⶨ���__________���ƫ�ߡ���ƫ�͡�������Ϊʵ��װ�û�����һ�����Ե�ȱ���� ��
���𰸡���1������һ��������2NaHCO3![]() Na2CO3��H2O��CO2�������γ�����������0.1gΪֹ
Na2CO3��H2O��CO2�������γ�����������0.1gΪֹ
��2����������HCO3-+OH-+Ba2+==BaCO3��+H2O��ȡ����С�ձ��е��ϲ���Һ���Թ��У��μ�һ��Ba��OH��2��Һ���۲��Ƿ��а�ɫ�������ɣ���û���������ȫ������������Ҳ��������������
��3����������Ũ�����ֹ�����е�ˮ������CO2����Cװ�ã�70.4%
ƫ����ȱ��һ��A��Bװ���ڵ�CO2�������ϵ�Cװ���е�װ������������������
��������
�����������1�� �������չ�������������������a����������������ʱ̼�����Ʒֽ�Ϊ̼���ơ�������̼��ˮ������������Ӧ�Ļ�ѧ����ʽΪ2NaHCO3![]() Na2CO3��H2O��CO2�������γ�����������0.1gΪֹ��֤��ʵ����NaHCO3�ֽ���ȫ����NaHCO3������Ba��OH��2��Һ��Ӧ�����ӷ���ʽHCO3-+OH-+Ba2+==BaCO3��+H2O��ʵ�����жϳ����Ƿ���ȫ�ķ�����ȡ����С�ձ��е��ϲ���Һ���Թ��У��μ�һ��Ba��OH��2��Һ���۲��Ƿ��а�ɫ�������ɣ���û���������ȫ�����˲����У����ձ���©���⣬��Ҫ����������������3������3���������ᷴӦ���ɶ�����̼����������NaHCO3�����������������ü�ʯ�����ն�����̼ǰҪ��Ũ�����������и��Bװ������ʢ�Լ���Ũ���Dװ�õ���������ֹ�����е�ˮ������CO2����Cװ��������Ʒ��̼���ơ�̼�����Ƶ����ʵ����ֱ���xmol��ymol��
Na2CO3��H2O��CO2�������γ�����������0.1gΪֹ��֤��ʵ����NaHCO3�ֽ���ȫ����NaHCO3������Ba��OH��2��Һ��Ӧ�����ӷ���ʽHCO3-+OH-+Ba2+==BaCO3��+H2O��ʵ�����жϳ����Ƿ���ȫ�ķ�����ȡ����С�ձ��е��ϲ���Һ���Թ��У��μ�һ��Ba��OH��2��Һ���۲��Ƿ��а�ɫ�������ɣ���û���������ȫ�����˲����У����ձ���©���⣬��Ҫ����������������3������3���������ᷴӦ���ɶ�����̼����������NaHCO3�����������������ü�ʯ�����ն�����̼ǰҪ��Ũ�����������и��Bװ������ʢ�Լ���Ũ���Dװ�õ���������ֹ�����е�ˮ������CO2����Cװ��������Ʒ��̼���ơ�̼�����Ƶ����ʵ����ֱ���xmol��ymol��![]() ��
��![]() ����Ʒ��NaHCO3����������Ϊ0.15��84��17.9��100%=70.4%��A��Bװ���ڵ�CO2��������ȫ�����ϵ�Cװ������ʹ�����ⶨ���ƫ����
����Ʒ��NaHCO3����������Ϊ0.15��84��17.9��100%=70.4%��A��Bװ���ڵ�CO2��������ȫ�����ϵ�Cװ������ʹ�����ⶨ���ƫ����

 ��ǰ����ϵ�д�
��ǰ����ϵ�д�