��Ŀ����
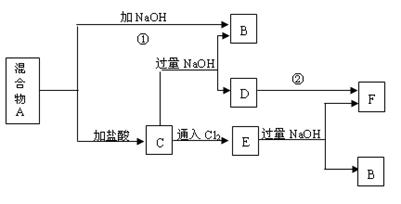
��12�֣����н�������A��B��C������ס��ҡ���������D��E��F��G��H������֮���ܷ������·�Ӧ��ͼ����Щ��Ӧ�IJ���ͷ�Ӧ������û��ȫ���������
�����������Ϣ�ش��������⣺
��1��д���������ʵĻ�ѧʽ���ף� ���ң� ��F�� ��H ��
��2������B������ǿ��������ǿ�Ӧ���ý���B�ǣ� ���ѧʽ��
����C�������������KSCN��Һ��Ѫ��ɫ���ý���C�ǣ� ���ѧʽ��
��3����Ӧ�۵Ļ�ѧ����ʽ ��
��4����Ӧ�ݵ����ӷ���ʽ ��

�����������Ϣ�ش��������⣺
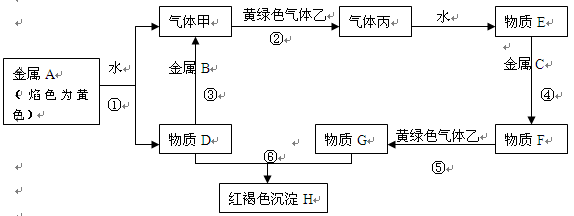
��1��д���������ʵĻ�ѧʽ���ף� ���ң� ��F�� ��H ��
��2������B������ǿ��������ǿ�Ӧ���ý���B�ǣ� ���ѧʽ��
����C�������������KSCN��Һ��Ѫ��ɫ���ý���C�ǣ� ���ѧʽ��
��3����Ӧ�۵Ļ�ѧ����ʽ ��
��4����Ӧ�ݵ����ӷ���ʽ ��
��1���ף� H2 ���ң� Cl2 ��F�� FeCl2 ��H Fe(OH) 3 ��
��2������B�ǣ� Al ���ѧʽ��
����C�ǣ� Fe ���ѧʽ��
��3����Ӧ�۵Ļ�ѧ����ʽ 2Al+2H2O+2NaOH= 2NaAlO2+3H2�� ��
��4����Ӧ�ݵ����ӷ���ʽ 2Fe 2+ +Cl2 = 2Fe3+ +2Cl- ��
��2������B�ǣ� Al ���ѧʽ��
����C�ǣ� Fe ���ѧʽ��
��3����Ӧ�۵Ļ�ѧ����ʽ 2Al+2H2O+2NaOH= 2NaAlO2+3H2�� ��
��4����Ӧ�ݵ����ӷ���ʽ 2Fe 2+ +Cl2 = 2Fe3+ +2Cl- ��
��

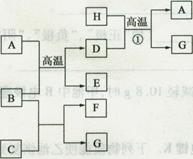
��ϰ��ϵ�д�
 �������ͬ����ϰϵ�д�
�������ͬ����ϰϵ�д�
�����Ŀ

 B
B D
D Bת���Ļ�ѧ����ʽ�� ��
Bת���Ļ�ѧ����ʽ�� ��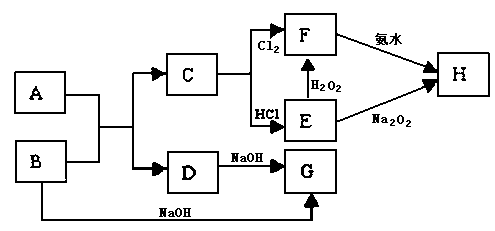
 �����ʹ���������ӷ���ʽ�� ��
�����ʹ���������ӷ���ʽ�� ��