��Ŀ����
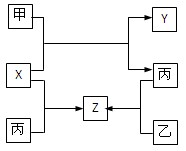
��10�֣�A��B��C��D��E��F�������ʵ�ת����ϵ��ͼ��ʾ(��Ӧ�����Ͳ��ֲ���δ���)��
(1)��AΪ�����ڽ������ʣ�DΪ�����ڷǽ������ʣ�������Ԫ�ص�ԭ������A��D��2��������Ԫ�ص�ԭ������������D��A��2����F��Ũ��Һ��A��D��Ӧ���к���ɫ�������ɣ���A��ԭ�ӽṹʾ��ͼΪ________����Ӧ�ܵĻ�ѧ����ʽΪ______________________________��
(2)��A�dz����ı�۽����ĵ��ʣ�D��F����̬���ʣ��ҷ�Ӧ����ˮ��Һ�н��С���Ӧ��Ҳ��ˮ��Һ�н��У������ӷ���ʽ��__________________________________��
�õ���ʽ��ʾ������B���γɹ��̣�_______________________________��
(3)��A��D��F���Ƕ����ڷǽ���Ԫ�ص��ʣ���A��D����Ԫ��ͬ���壬A��F����Ԫ��
ͬ���ڣ���Ӧ�ٵĻ�ѧ����ʽΪ___________________________________��
��3��ͬ��ǽ���Ԫ��֮����û���һ���ǵڢ�A���ڢ�A������A��

(1)��AΪ�����ڽ������ʣ�DΪ�����ڷǽ������ʣ�������Ԫ�ص�ԭ������A��D��2��������Ԫ�ص�ԭ������������D��A��2����F��Ũ��Һ��A��D��Ӧ���к���ɫ�������ɣ���A��ԭ�ӽṹʾ��ͼΪ________����Ӧ�ܵĻ�ѧ����ʽΪ______________________________��
(2)��A�dz����ı�۽����ĵ��ʣ�D��F����̬���ʣ��ҷ�Ӧ����ˮ��Һ�н��С���Ӧ��Ҳ��ˮ��Һ�н��У������ӷ���ʽ��__________________________________��
�õ���ʽ��ʾ������B���γɹ��̣�_______________________________��
(3)��A��D��F���Ƕ����ڷǽ���Ԫ�ص��ʣ���A��D����Ԫ��ͬ���壬A��F����Ԫ��
ͬ���ڣ���Ӧ�ٵĻ�ѧ����ʽΪ___________________________________��
��3��ͬ��ǽ���Ԫ��֮����û���һ���ǵڢ�A���ڢ�A������A��
(1) ��2�֣�
��2�֣�
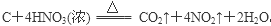
(2)2Fe2����Cl2== 2Fe3����2Cl����2�֣�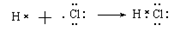
(3) ��2�֣�
��2�֣�
 ��2�֣�
��2�֣�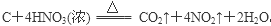
(2)2Fe2����Cl2== 2Fe3����2Cl����2�֣�
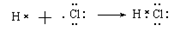
(3)
 ��2�֣�
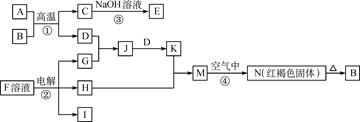
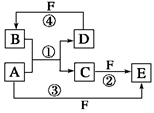
��2�֣���1����Ӧ�������û���Ӧ����Ϊ�����û��ǽ����ķ�Ӧ������Ϊ����Ԫ�ص�ԭ������A��D��2��������Ԫ�ص�ԭ������������D��A��2������˷���������ֻ��þ�û�̼����þ�ڶ�����̼��ȼ�����ɵ���̼������ɫ�����Ƕ�������������F�����ᡣ��Ӧ�ܵĻ�ѧ����ʽΪ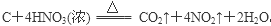 ��
��
��2�������ı�۽�������������ת��ͼ���Եó�ABCDER�ֱ����������ᣬ�Ȼ��������������Ȼ�������������Ӧ�ڵ����ӷ���ʽ��2Fe2����Cl2== 2Fe3����2Cl����B���Ȼ��⣬���ڹ��ۻ�������γɹ���Ϊ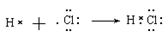 ��
��
��3��ͬ��ǽ���Ԫ��֮����û���һ���ǵڢ�A���ڢ�A������A������ת��ͼ����
���Կ���ACE�о�����AԪ�أ���Ϊ��۵ķǽ���Ԫ�ء����Է�����������C��CO��CO2�����Է�Ӧ�ٵĻ�ѧ����ʽΪ ��
��
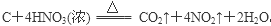 ��
����2�������ı�۽�������������ת��ͼ���Եó�ABCDER�ֱ����������ᣬ�Ȼ��������������Ȼ�������������Ӧ�ڵ����ӷ���ʽ��2Fe2����Cl2== 2Fe3����2Cl����B���Ȼ��⣬���ڹ��ۻ�������γɹ���Ϊ
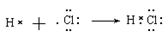 ��
����3��ͬ��ǽ���Ԫ��֮����û���һ���ǵڢ�A���ڢ�A������A������ת��ͼ����
���Կ���ACE�о�����AԪ�أ���Ϊ��۵ķǽ���Ԫ�ء����Է�����������C��CO��CO2�����Է�Ӧ�ٵĻ�ѧ����ʽΪ
 ��
��
��ϰ��ϵ�д�
�����Ŀ
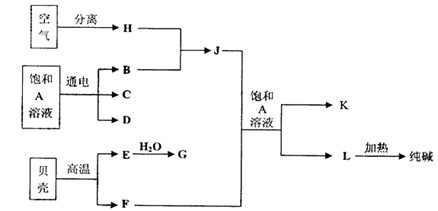
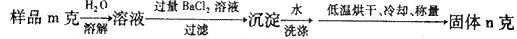
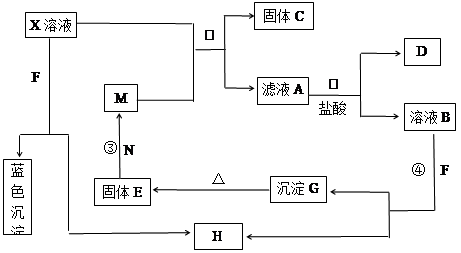


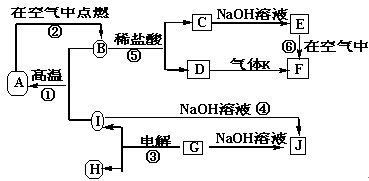
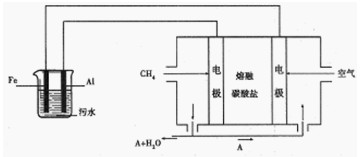
 ��
��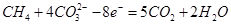 ��
��